Chapter 16: PROBLEM 16.24 (page 627)
Question: Draw the product of each Diels–Alder reaction.
a.
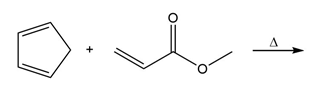
b.
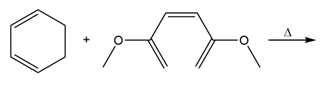
Short Answer
Answer
a.
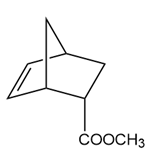
b.
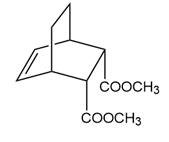
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.24 (page 627)
Question: Draw the product of each Diels–Alder reaction.
a.

b.

Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question. Draw the structure consistent with each description.
a. (2E,4E)-octa-2,4-diene in the s-trans conformation
b. (3E,5Z)-nona-3,5-diene in the s-cis conformation
c. (3Z,5Z)-4,5-dimethyldeca-3,5-diene. Draw both the s-cis and s-trans conformations.
Question: What diene and dienophile are needed to prepare each Diels–Alder product?
a.
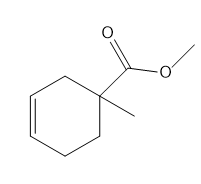
b.
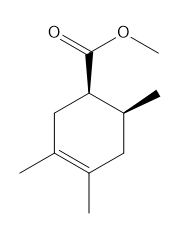
c.
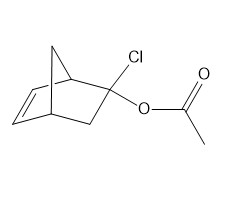
d.
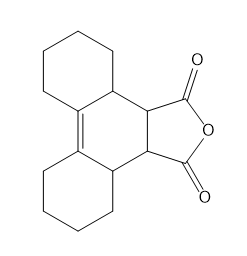
Question: Use resonance theory and the Hammond postulate to explain why 3-chloroprop-1-ene is more reactive than 1-chloropropane in SN1 reactions.
Question: Draw the product formed when each diene and dienophile react in a Diels–Alder reaction.
a.
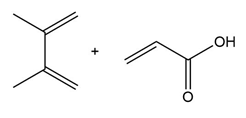
b.
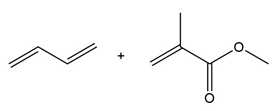
c.
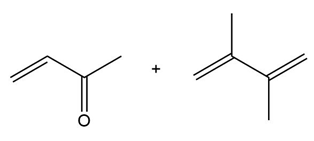
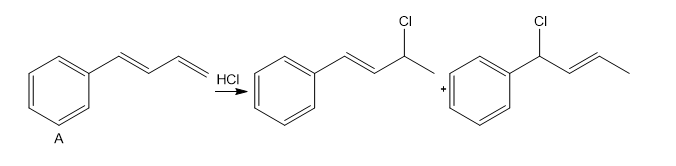
Question: Explain, with reference to the mechanism, why addition of one equivalent of HCl to diene A forms only two products of electrophilic addition, even though four constitutional isomers are possible.
What do you think about this solution?
We value your feedback to improve our textbook solutions.