Chapter 16: Conjugation, Resonance, and Dienes
PROBLEM 16.38
Question: Draw all possible stereoisomers of hepta-2,4-diene and label each double bond as E or Z.
PROBLEM 16.39
Question: Label each pair of compounds as stereoisomers or conformations.
a.
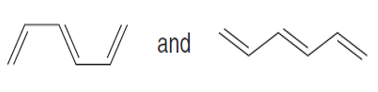
b.
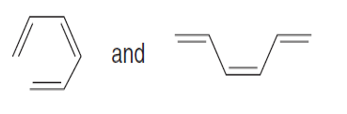
c.
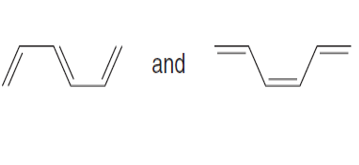
PROBLEM 16.4
Question: Use resonance theory and the Hammond postulate to explain why 3-chloroprop-1-ene is more reactive than 1-chloropropane in SN1 reactions.
PROBLEM 16.40
Question: Rank the following dienes in order of increasing heat of hydrogenation.
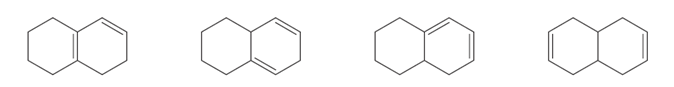
PROBLEM 16.41
Question: Draw the products formed when each compound is treated with one equivalent of HBr.
a.
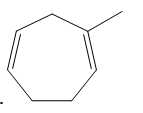
b.
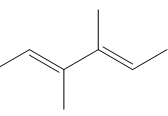
c.
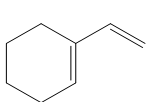
PROBLEM 16.42
Question: Ignoring stereoisomers, draw all products that form by addition of HBr to (E)-hexa-1,3,5-triene.
PROBLEM 16.43
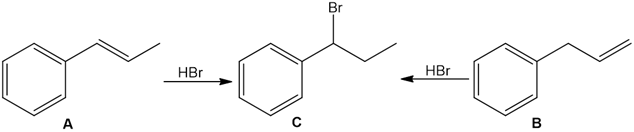
Question: Treatment of alkenes A and B with HBr gives the same alkyl halide C. Draw a mechanism for each reaction, including all reasonable resonance structures for any intermediate.
PROBLEM 16.44
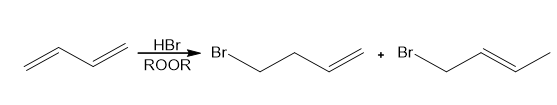
Question: Draw a stepwise mechanism for the following reaction.
PROBLEM 16.45
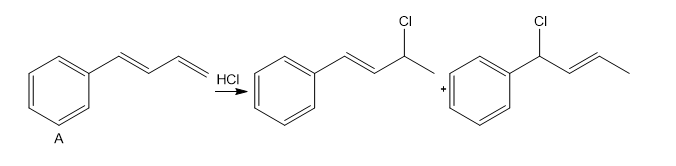
Question: Addition of HCl to alkene X forms two alkyl halides Y and Z.
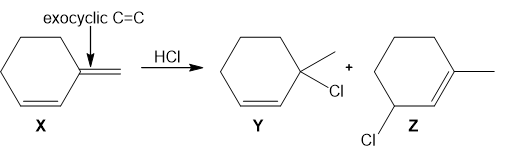
a. Label Y and Z as a 1,2-addition product or a 1,4-addition product.
b. Label Y and Z as the kinetic or thermodynamic product and explain why.
c. Explain why addition of HCl occurs at the indicated C=C(called an exocyclic double bond), rather than the other C=C (called an endocyclic double bond).
PROBLEM 16.46
Question: Explain, with reference to the mechanism, why addition of one equivalent of HCl to diene A forms only two products of electrophilic addition, even though four constitutional isomers are possible.