Chapter 26: Q 21. (page 1069)
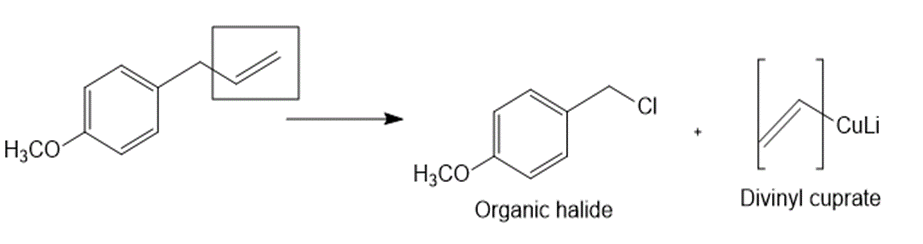
What organic halide is needed to convert lithium divinylcuprate to each compound?
a.
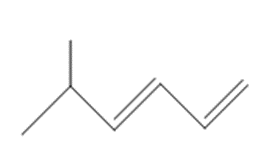
b.
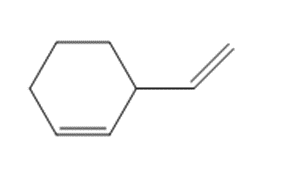
c.
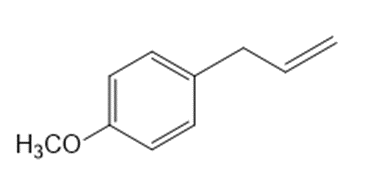
Short Answer
Answer
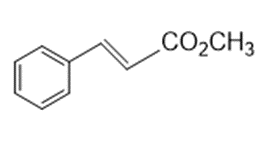
a.
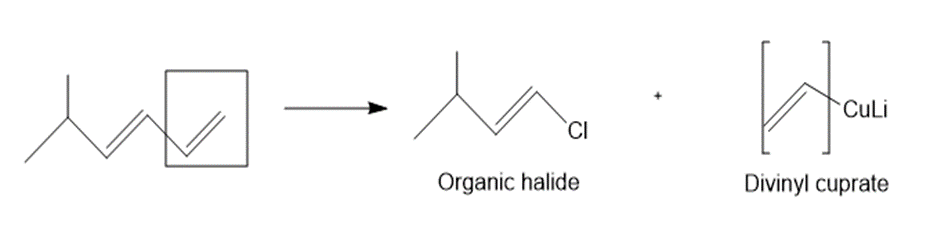
b.

c.
Learning Materials
Features
Discover
Chapter 26: Q 21. (page 1069)
What organic halide is needed to convert lithium divinylcuprate to each compound?
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Treatment of cyclohexene with and Zn(Cu) forms two stereoisomers of molecular formula . Draw their structures and explain why two compounds are formed.
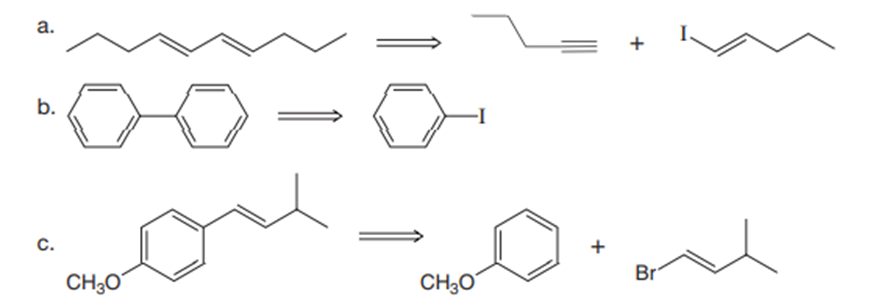
Devise a synthesis of each of the following compounds. Besides inorganic reagents, you may use hydrocarbons and halides having C’s, and as starting materials. Each synthesis must use at least one of the carbon–carbon bond-forming reactions in this chapter.
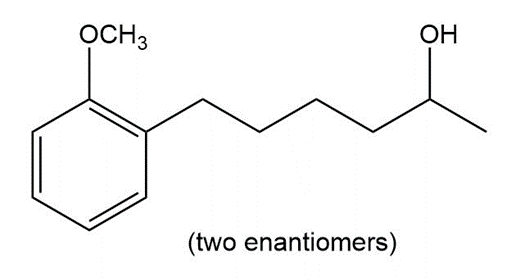
b.
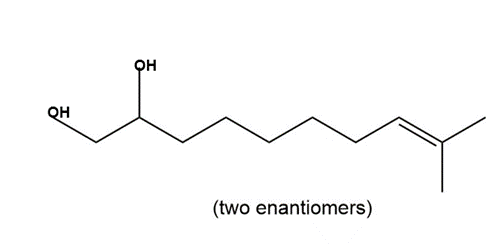
c.
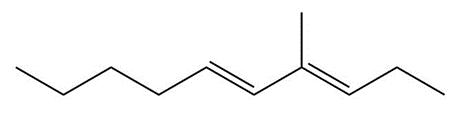
d.
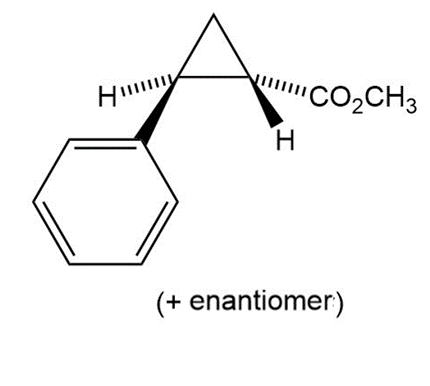
Synthesize each compound from the given starting materials.
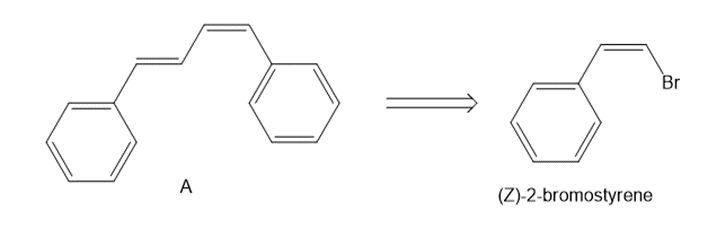
Devise a synthesis of diene A from (Z)-2-bromostyrene as the only organic starting material. Use a Suzuki reaction in one step of the synthesis.
Devise a synthesis of each compound from benzene. You may also use any organic compounds having four carbons or fewer, and any required inorganic reagents.
a.
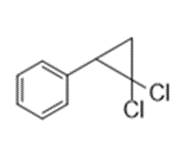
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.