Chapter 26: Q 19. (page 1068)
What product is formed by ring-closing metathesis of each compound?
a.
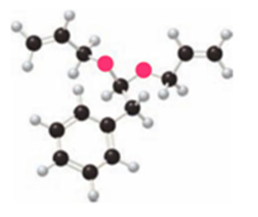
b.
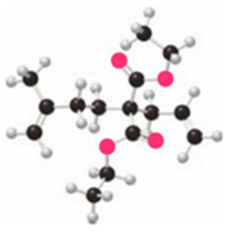
Short Answer
Answer
a.
b.
Learning Materials
Features
Discover
Chapter 26: Q 19. (page 1068)
What product is formed by ring-closing metathesis of each compound?
a.

b.

Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
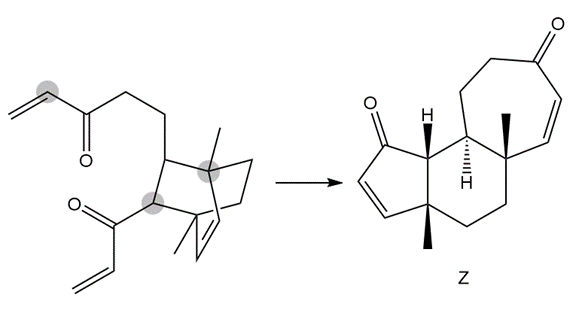
The following conversion, carried out in the presence of Grubbs catalyst and ethylene gas, involves a cascade of metathesis reactions. Draw a reaction sequence that illustrates how the reactant is converted to the product Z, and indicate where each labeled atom in the reactant ends up in Z.
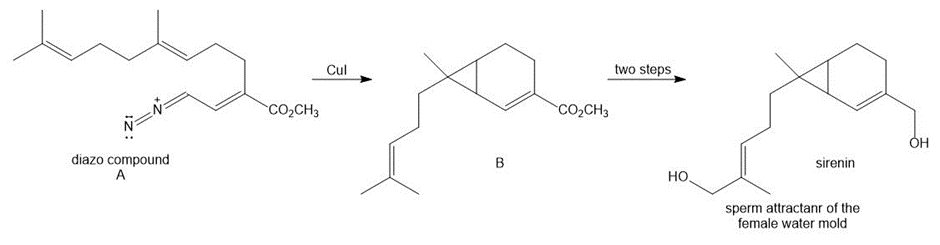
Although diazomethane ( ) is often not a useful reagent for preparing cyclopropanes,other diazo compounds give good yields of more complex cyclopropanes. Draw a stepwisemechanism for the conversion of diazo compound A to B, an intermediate in the synthesis ofsirenin, the sperm attractant produced by the female gametes of the water mold Allomyces.
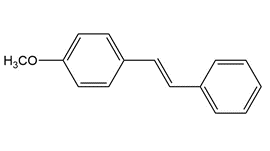
Devise a synthesis of each compound using a Heck reaction as one step. You may use benzene, , organic alcohols having two carbons or fewer and any required inorganic reagents.
a.
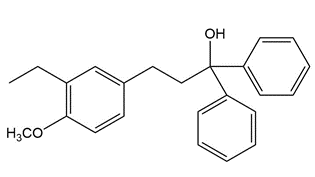
b.
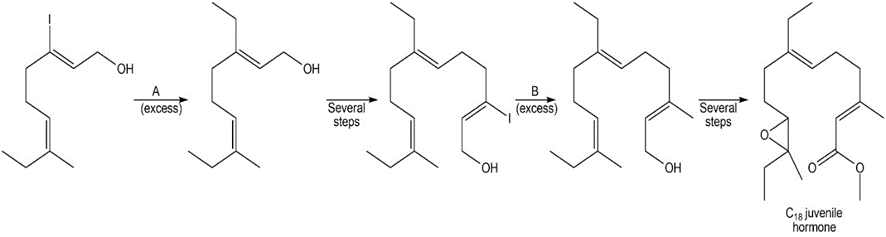
Identify reagents A and B in the following reaction scheme. This synthetic sequence was used to prepare the C18 juvenile hormone (Figure 20.6).
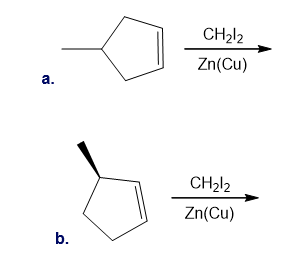
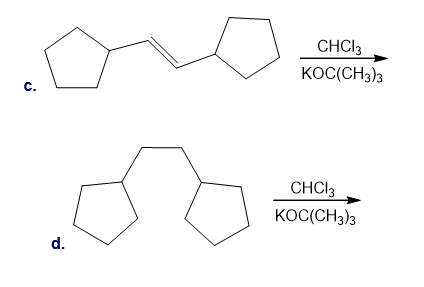
Draw the products (including stereoisomers) formed in each reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.