Chapter 17: Q.18. (page 641)
Question: Assuming the rings are planar, which ions are aromatic?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 17: Q.18. (page 641)
Question: Assuming the rings are planar, which ions are aromatic?
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: AZT was the first drug approved to treat HIV, the virus that causes AIDS. Explain why the six-membered ring of AZT is aromatic.

Question: Which of the diethylbenzene isomers (ortho, meta, or para) corresponds to each set of NMR spectral data?
[A] NMR signals: 16, 29, 125, 127.5, 128.4, and 144 ppm
[B] NMR signals: 15, 26, 126, 128, and 142 ppm
[C] NMR signals: 16, 29, 128, and 141 ppm
Question: What is the structure of a compound of molecular formula that shows a strong IR absorption at 3150–2850 role="math" localid="1648710122642" and gives the following 1 H NMR absorptions: 1.4 (triplet, 6 H), 4.0 (quartet, 4 H), and 6.8 (singlet, 4 H) ppm
Question:
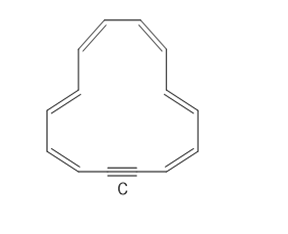
a. How many electrons does C contain?
b. How many electrons are delocalized in the ring?
c. Explain why C is aromatic.
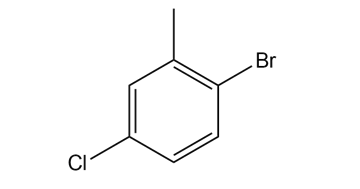
Give the IUPAC name for each compound.
(a)
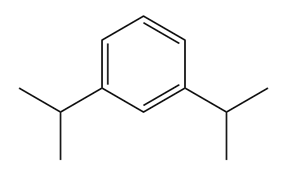
(b)
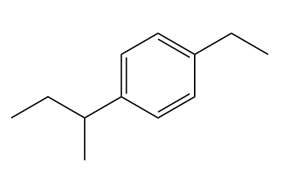
(c)
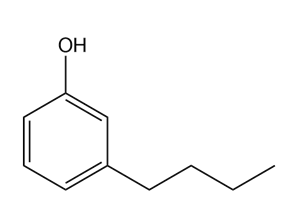
(d)
What do you think about this solution?
We value your feedback to improve our textbook solutions.