Chapter 29: 65P (page 1197)
Draw the mechanism for the reaction that removes an Fmoc group from an amino acid under the following conditions:
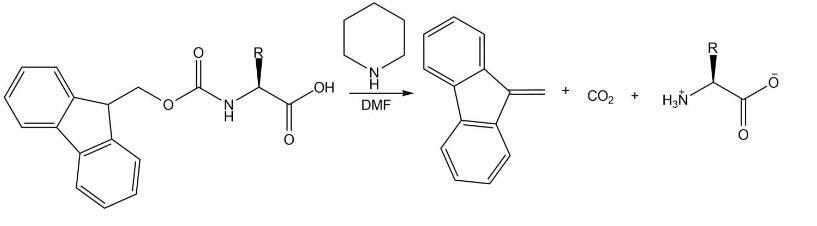
Short Answer

Learning Materials
Features
Discover
Chapter 29: 65P (page 1197)
Draw the mechanism for the reaction that removes an Fmoc group from an amino acid under the following conditions:


All the tools & learning materials you need for study success - in one app.
Get started for free
Consider the decapeptide angiotensin I.

The larger polypeptide is angiotensin II, a hormone that narrows blood vessels and increases blood pressure. Give the amino acid sequence of angiotensin II using threeletter abbreviations. ACE inhibitors are drugs that lower blood pressure by inhibiting the ACE enzyme (Problem 5.15).
Besides the Boc and Fmoc protecting groups used in peptide synthesis, amines can also be protected by reaction with benzyl chloroformate (). Draw the structure of the product formed by reaction of alanine with benzyl chloroformate
Question: Identify the functional groups in each molecule. Classify each alcohol, alkyl halide, amide, and amine as 1°, 2°, or 3°
a.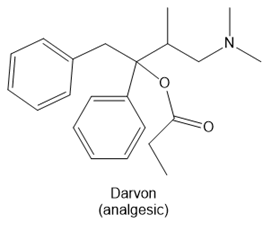
b.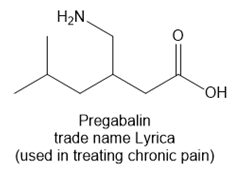
c. 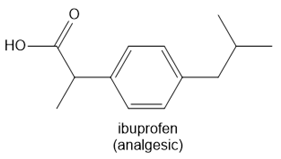
d. 
e. 
f. 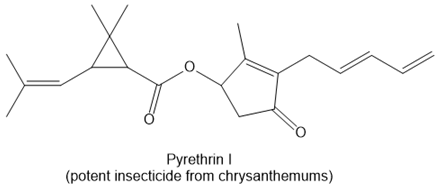
What aldehyde is needed to synthesize each amino acid by the Strecker synthesis?
(a) Valine;
(b) Leucine;
(c) Phenylalanine.
Draw the structure of the N-phenylthiohydantoin formed by initial Edman degradation of each peptide: (a) Ala-Gly-Phe-Phe; (b) Val-Ile-Tyr.
What do you think about this solution?
We value your feedback to improve our textbook solutions.