Chapter 29: Q19. (page 1170)
Draw the structure of the N-phenylthiohydantoin formed by initial Edman degradation of each peptide: (a) Ala-Gly-Phe-Phe; (b) Val-Ile-Tyr.
Short Answer
a.
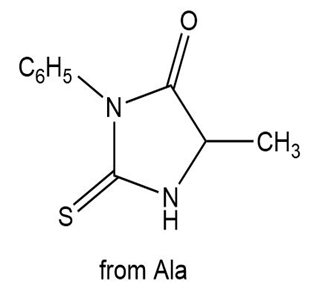
b.
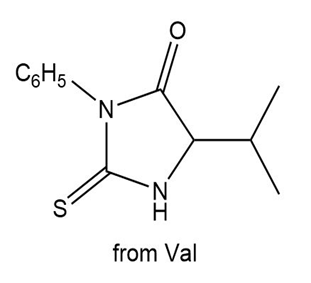
Learning Materials
Features
Discover
Chapter 29: Q19. (page 1170)
Draw the structure of the N-phenylthiohydantoin formed by initial Edman degradation of each peptide: (a) Ala-Gly-Phe-Phe; (b) Val-Ile-Tyr.
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
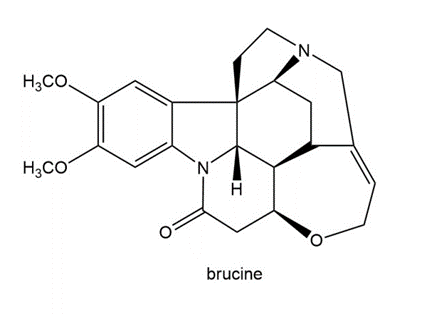
Brucine is a poisonous alkaloid obtained from Strychnosnux vomica, a tree that grows in India, Sri Lanka, and northern Australia. Write out a resolution scheme similar to the one given in Section 29.3A, which shows how a racemic mixture of phenylalanine can be resolved using brucine
Draw the amino acids and peptide fragments formed when the decapeptide A-P-F-L-K-W-S-G-R-G is treated with each reagent or enzyme: (a) chymotrypsin; (b) trypsin; (c) carboxypeptidase; (d)
Explain why the of the group of an -amino acid is lower than the of the ammonium ion derived from a amine . For example, the of therole="math" localid="1648617659352" group of alanine is 9.87 but the of is 10.63.
Draw the structure for each peptide:
(a) Phe–Ala;
(b) Gly–Gln;
(c) Lys–Gly;
(d) R-H
Draw the structure of each peptide. Label the N-terminal and C-terminal amino acids and all amide bonds.
a. Val–Glu
b. Gly–His–Leu
c. M–A–T–T
What do you think about this solution?
We value your feedback to improve our textbook solutions.