Chapter 25: PROBLEM 25.14 (page 1012)
Question: Enalapril, a drug used to treat hypertension, is prepared from compounds D and E by reductive amination. What is the structure of enalapril?
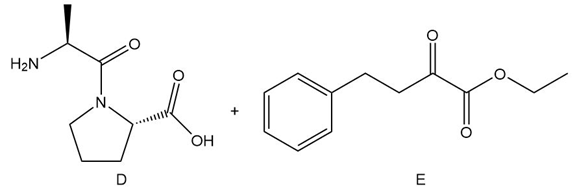
Short Answer
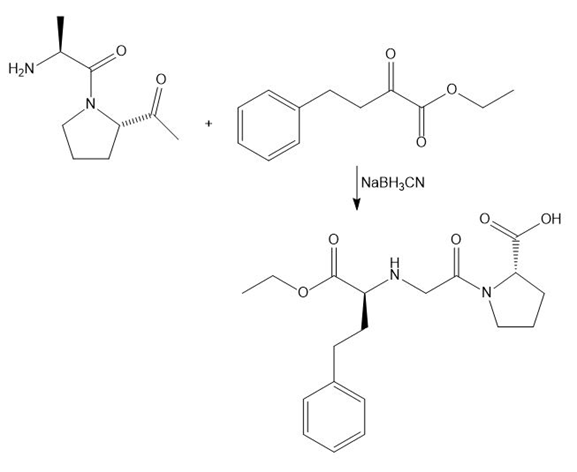
Answer
Learning Materials
Features
Discover
Chapter 25: PROBLEM 25.14 (page 1012)
Question: Enalapril, a drug used to treat hypertension, is prepared from compounds D and E by reductive amination. What is the structure of enalapril?

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
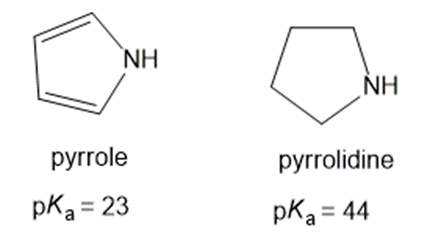
Why is pyrrole more acidic than pyrrolidine?
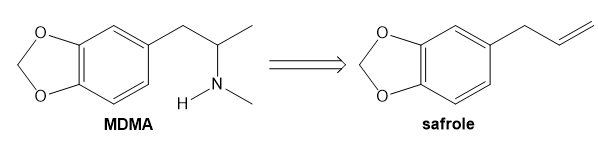
Question: Safrole, which is isolated from sassafras (Problem 21.33), can be converted to the illegal stimulant MDMA (3,4-methylenedioxymethamphetamine, “Ecstasy”) by a variety of methods. (a) Devise a synthesis that begins with safrole and uses a nucleophilic substitution reaction to introduce the amine. (b) Devise a synthesis that begins with safrole and uses reductive amination to introduce the amine.
Question: Arrange the compounds in order of increasing boiling point.
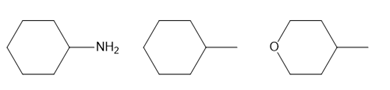
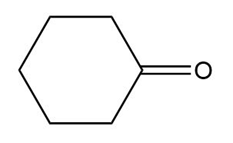
Question: Draw the products formed when each carbonyl compound reacts with the following amines:
1. CH3CH2CH2NH2
2.(CH3CH2)2NH
a.
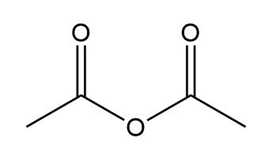
b.
c.
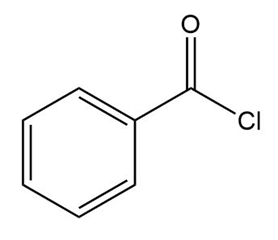
Question: Draw the major product formed in each reaction.
a.
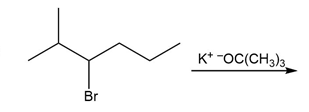
b.
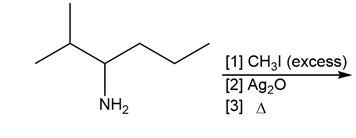
c.
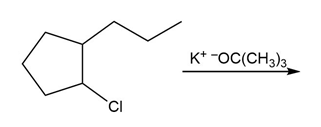
d.
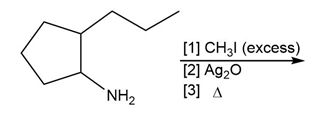
What do you think about this solution?
We value your feedback to improve our textbook solutions.