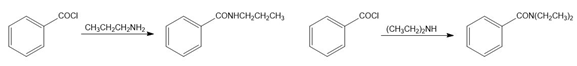
Chapter 25: PROBLEM 25.24 (page 1023)
Question: Draw the products formed when each carbonyl compound reacts with the following amines:
1. CH3CH2CH2NH2
2.(CH3CH2)2NH
a.
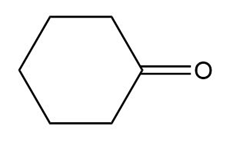
b.
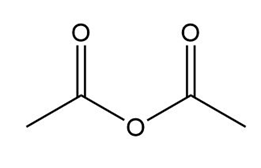
c.
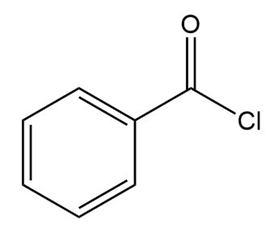
Short Answer
Answer
a.

b.
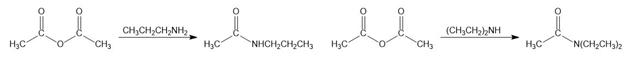
c.
Learning Materials
Features
Discover
Chapter 25: PROBLEM 25.24 (page 1023)
Question: Draw the products formed when each carbonyl compound reacts with the following amines:
1. CH3CH2CH2NH2
2.(CH3CH2)2NH
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
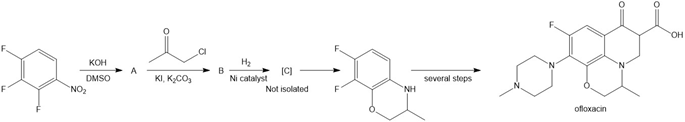
Question: Identify the intermediates (A–C) in the following reaction sequence, which was used to prepare racemic ofloxacin. One enantiomer of the product, levofloxacin, is an antibiotic used to treat severe bacterial infections that have not responded to other drugs.
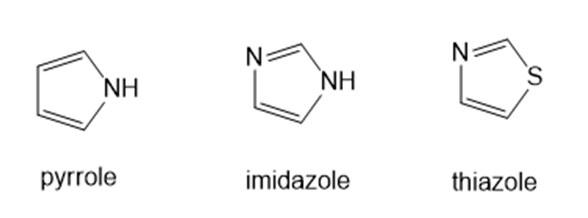
Rank the following compounds in order of increasing basicity and explain the order you choose.
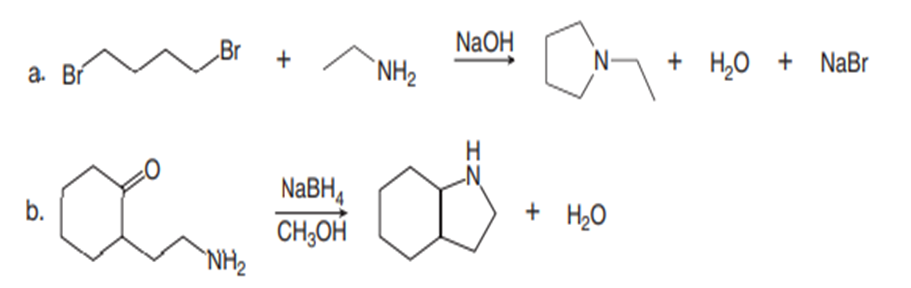
Draw a stepwise mechanism for each reaction.
Question: Which amines cannot be prepared by a Gabriel synthesis? Explain your choices.
a.
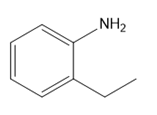
b.
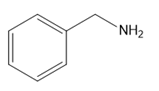
c.
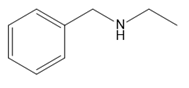
d.
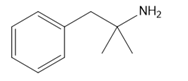
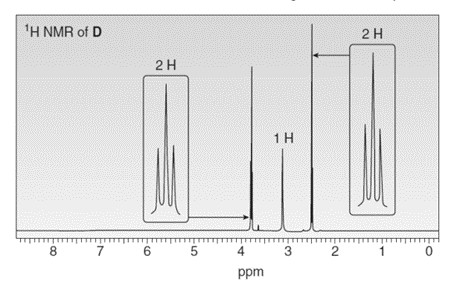
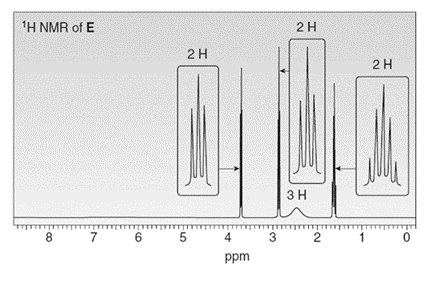
Treatment of compound D with followed by forms compound E. D shows a molecular ion in its mass spectrum at m/z= 71 and IR absorptions at and . E shows a molecular ion in its mass spectrum at m/z= 75 and IR absorptions at and . Propose structures for D and E from these data and the given 1H NMR spectra.
What do you think about this solution?
We value your feedback to improve our textbook solutions.