Chapter 25: 76 (page 1048)
Draw the product Y of the following reaction sequence. Y was an intermediate in the remarkable synthesis of cyclooctatetraene by Wilstatter in 1911.
Short Answer
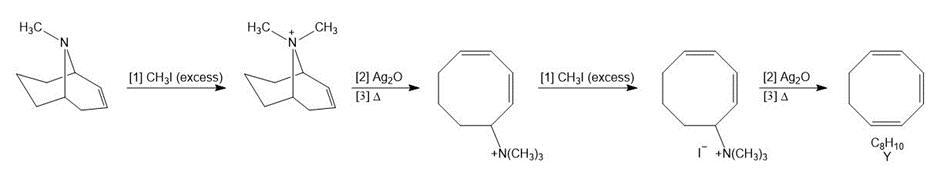
Mechanism of the given reaction
Learning Materials
Features
Discover
Chapter 25: 76 (page 1048)
Draw the product Y of the following reaction sequence. Y was an intermediate in the remarkable synthesis of cyclooctatetraene by Wilstatter in 1911.

Mechanism of the given reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
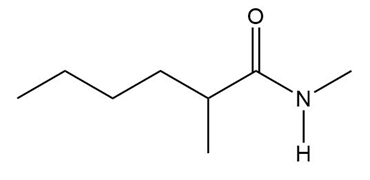
What amine is formed by reduction of each amide?
a.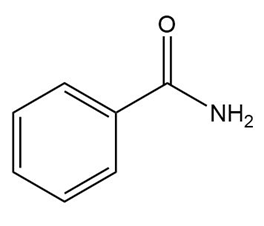
b.
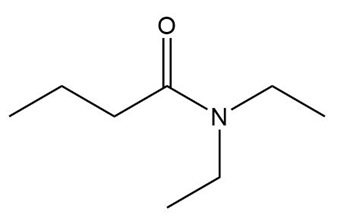
c.
Question: Synthesize each compound from benzene. Use a diazonium salt as one of the synthetic intermediates.
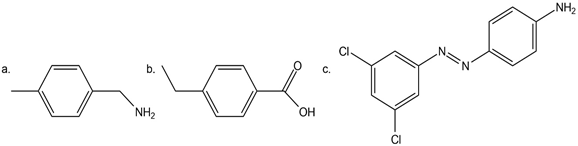
Devise a synthesis of each biologically active compound from benzene.
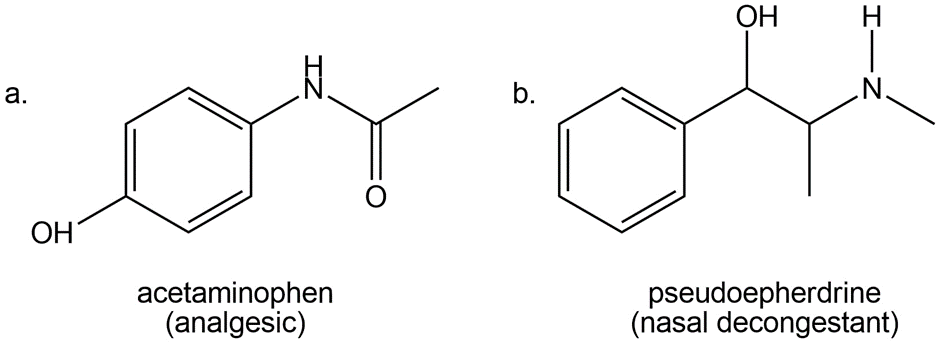
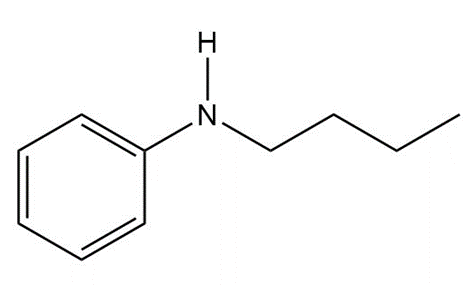
Question: Devise a synthesis of each compound from aniline (C6H5NH2)
a.
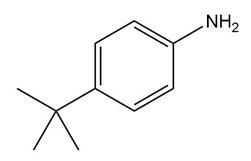
b.
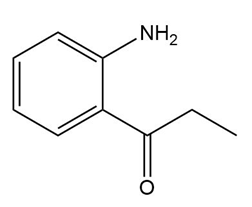
What carbonyl and nitrogen compounds are needed to make each compound by reductive amination? When more than one set of starting materials is possible, give all possible methods.
(a)
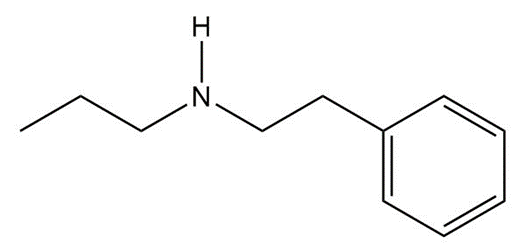
(b)
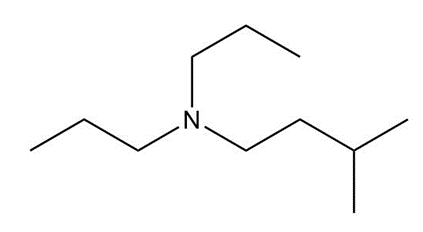
(c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.