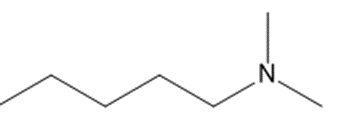
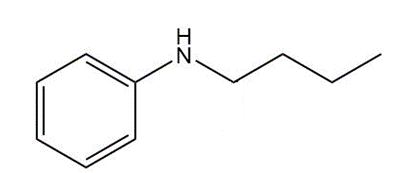
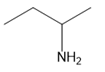
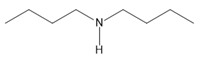
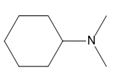
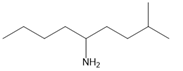
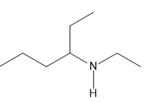
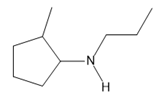
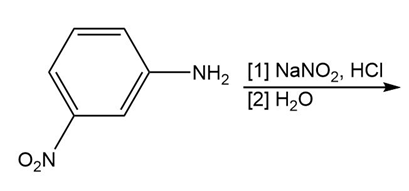
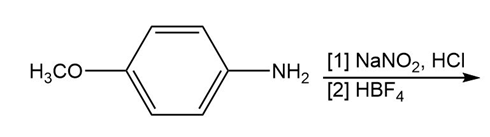
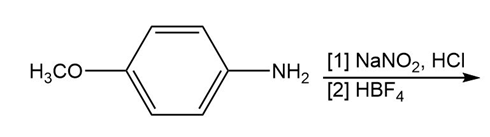
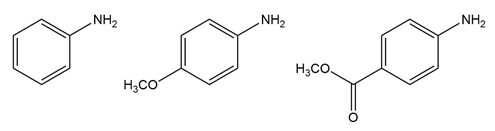
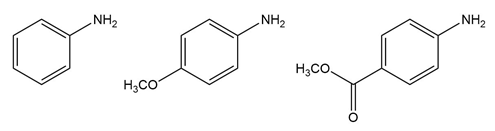
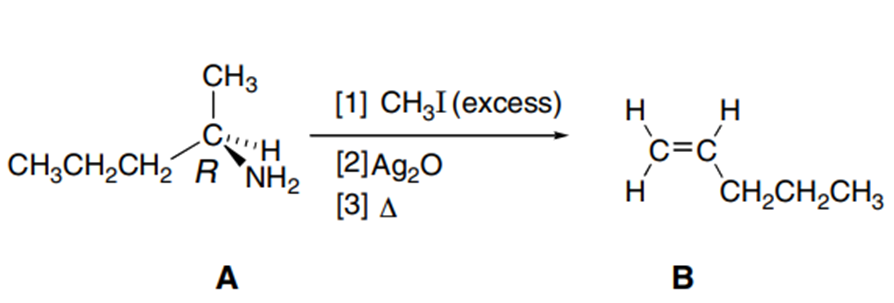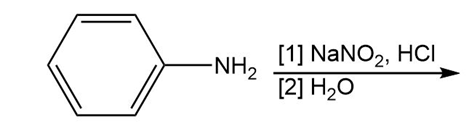Chapter 25: 60 (page 1045)
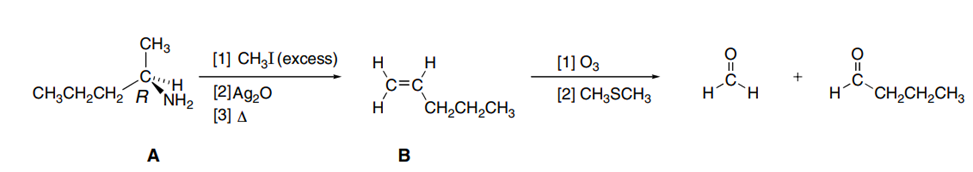
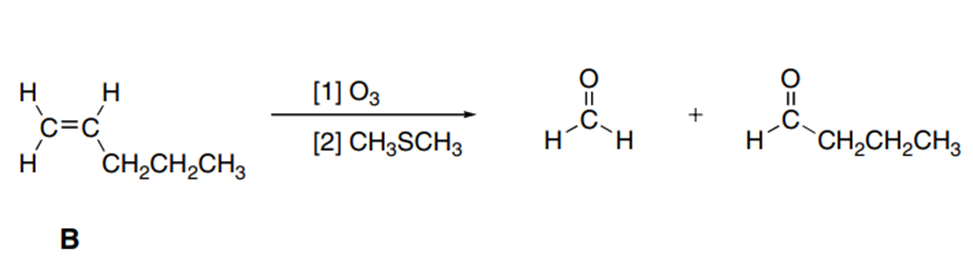
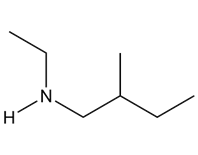
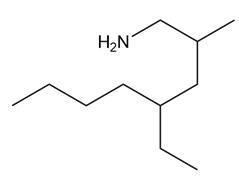
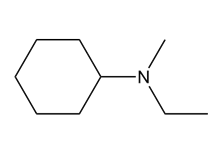
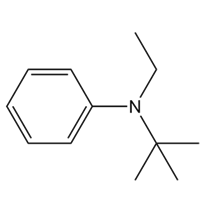
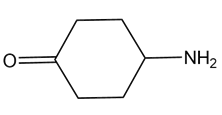
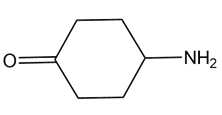
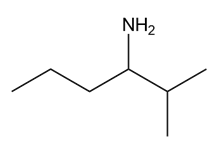
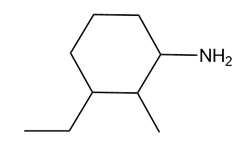
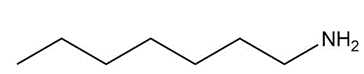
A chiral amine A having the R configuration undergoes Hofmann elimination to form an alkene B as the major product. B is oxidatively cleaved with ozone, followed by , to form and .What are the structures of A and B?
Short Answer