Chapter 7: PROBLEM 7.47 (page 291)
Question: Which of the following molecules contain a good leaving group?
a.
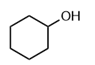
b.
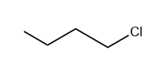
c.
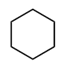
d.
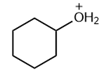
Short Answer
ANSWER
The answer is option (d).
Learning Materials
Features
Discover
Chapter 7: PROBLEM 7.47 (page 291)
Question: Which of the following molecules contain a good leaving group?
a.

b.

c.

d.

ANSWER
The answer is option (d).
All the tools & learning materials you need for study success - in one app.
Get started for free
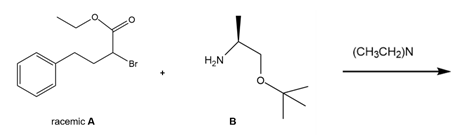
Question: Quinapril (trade name Accupril) is used to treat high blood pressure and congestive heart failure. One step in the synthesis of quinapril involves the reaction of the racemic alkyl bromide A with a single enantiomer of the amino ester B. (a) What two products are formed in this reaction? (b) Given the structure of quinapril, which one of these two products is needed to synthesize the drug?
 quinapril
quinapril
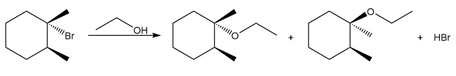
Question: Draw a stepwise, detailed mechanism for the following reaction. Use curved arrows to show the movement of electrons.
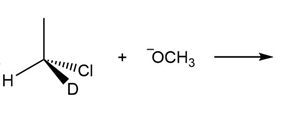
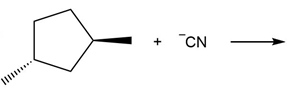
Question: Draw the products of each reaction and indicate the stereochemistry where appropriate.
a.
b.
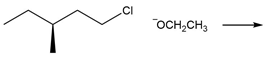
c.
Question: Give the structure corresponding to each name.
a. 3-chloro-2-methylhexane
b. 4-ethyl-5-iodo-2,2-dimethyloctane
c. cis-1,3-dichlorocyclopentane
d. 1,1,3-tribromocyclohexane
e. sec-butyl bromide
f. 6-ethyl-3-iodo-3,5-dimethylnonane
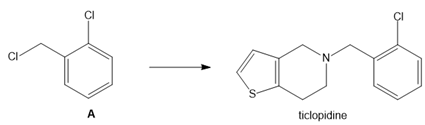
Question: What neutral nucleophile is needed to convert dihalide A to ticlopidine, an antiplatelet drug used to reduce the risk of strokes?
What do you think about this solution?
We value your feedback to improve our textbook solutions.