Chapter 7: PROBLEM 7.48 (page 291)
Question: Rank the species in each group in order of increasing leaving group ability.
a. Br-,Cl-,I-
b. NH3,H2S,H2O
Short Answer
Answer
a. Cl-< Br-<I-
b. NH3 < H2O <H2S
Learning Materials
Features
Discover
Chapter 7: PROBLEM 7.48 (page 291)
Question: Rank the species in each group in order of increasing leaving group ability.
a. Br-,Cl-,I-
b. NH3,H2S,H2O
Answer
a. Cl-< Br-<I-
b. NH3 < H2O <H2S
All the tools & learning materials you need for study success - in one app.
Get started for free
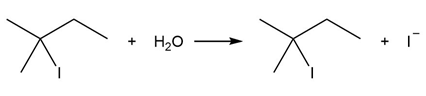
Question: Consider the following SN1 reaction.
Question: Give the structure corresponding to each name.
a. 3-chloro-2-methylhexane
b. 4-ethyl-5-iodo-2,2-dimethyloctane
c. cis-1,3-dichlorocyclopentane
d. 1,1,3-tribromocyclohexane
e. sec-butyl bromide
f. 6-ethyl-3-iodo-3,5-dimethylnonane
Question: Draw the eight constitutional isomers having the molecular formula C5H11Cl .
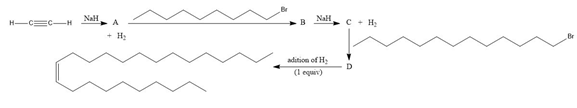
Question: Muscalure, the sex pheromone of the common housefly, can be prepared by a reaction sequence that uses two nucleophilic substitutions. Identify compounds A–D in the following synthesis of muscalure.
Question: Which compound in each pair undergoes a faster SN2 reaction?
a.
 and
and 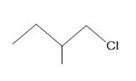
b.
 and
and 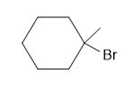
What do you think about this solution?
We value your feedback to improve our textbook solutions.