Chapter 7: PROPBLEM 7.9 (page 255)
Question: What neutral nucleophile is needed to convert dihalide A to ticlopidine, an antiplatelet drug used to reduce the risk of strokes?
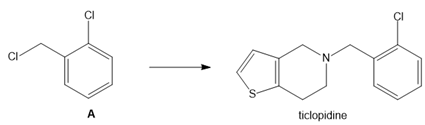
Short Answer
The structure of the neutral nucleophile is shown below:
Learning Materials
Features
Discover
Chapter 7: PROPBLEM 7.9 (page 255)
Question: What neutral nucleophile is needed to convert dihalide A to ticlopidine, an antiplatelet drug used to reduce the risk of strokes?

The structure of the neutral nucleophile is shown below:

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which compound in each pair has the higher boiling point?
a.
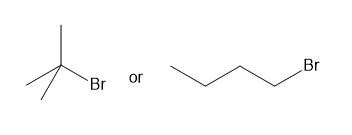
b.
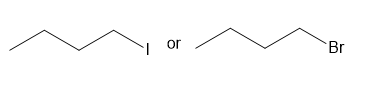
Question: Draw the products of each SN1 reaction and indicate the stereochemistry of any stereogenic centers.
a.
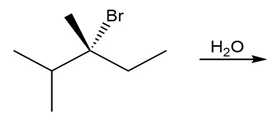
b.
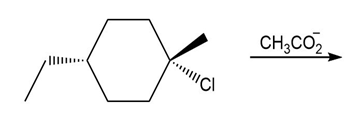
Question: Identify the stronger nucleophile in each pair of anions.
a. Br- or Cl- in a polar protic solvent.
b. OH-or Cl- in a polar aprotic solvent.
c. HS- or F- in a polar protic solvent.
Telfairine, a naturally occurring insecticide, and halomon, an antitumor agent, are two polyhalogenated compounds isolated from red algae. (a) Classify each halide bonded to an sp3 hybridized carbon as 1°, 2°, or 3°. (b) Label each halide as vinyl, allylic, or neither.

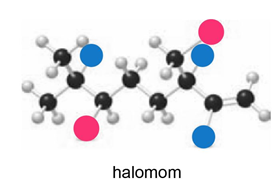
Question: What alkyl halide and nucleophile are needed to prepare each compound?
a.
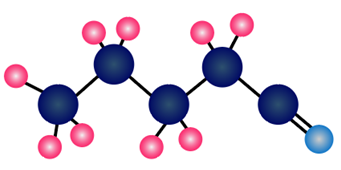
b.
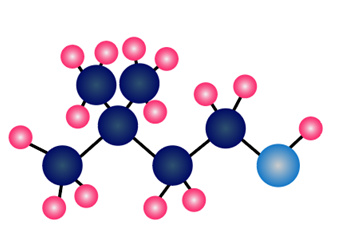
c.
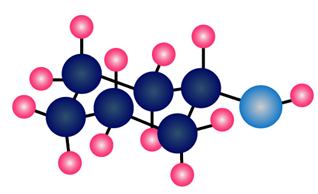
d.
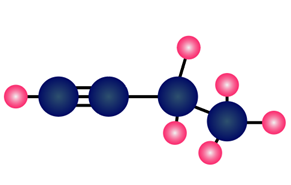
What do you think about this solution?
We value your feedback to improve our textbook solutions.