Chapter 7: PROBLEM 7.45 (page 290)
Question: Which compound in each pair has the higher boiling point?
a.
b.
Short Answer
ANSWER
a.
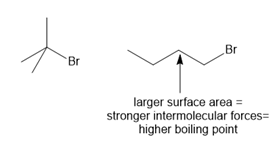
b.
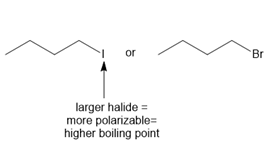
Learning Materials
Features
Discover
Chapter 7: PROBLEM 7.45 (page 290)
Question: Which compound in each pair has the higher boiling point?
a.

b.
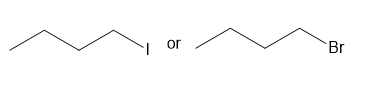
ANSWER
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which of the following nucleophilic substitution reactions will take place?
a.
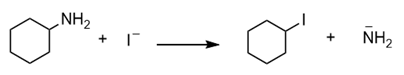
b.

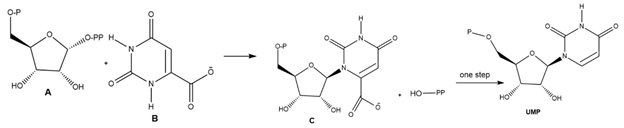
Question: Uridine monophosphate (UMP) is one of the four nucleotides that compose RNA, the nucleic acid that translates the genetic information of DNA into proteins needed by cells for proper function and development. A key step in the synthesis of UMP is the reaction of A with B to form C, which is then converted to UMP in one step. Draw a stepwise mechanism for this SN1 reaction.
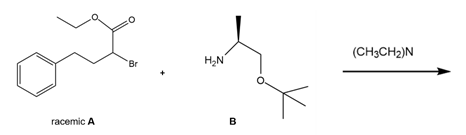
Question: Quinapril (trade name Accupril) is used to treat high blood pressure and congestive heart failure. One step in the synthesis of quinapril involves the reaction of the racemic alkyl bromide A with a single enantiomer of the amino ester B. (a) What two products are formed in this reaction? (b) Given the structure of quinapril, which one of these two products is needed to synthesize the drug?
 quinapril
quinapril
Question: Fill in the appropriate reagent or starting material in each of the following reactions:
a.
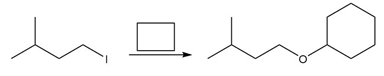
b.
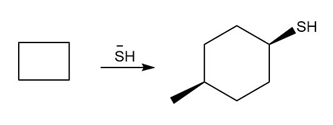
c.
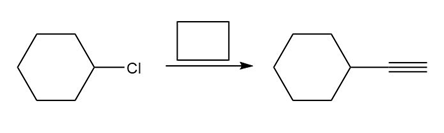
d.
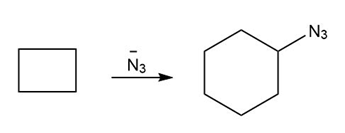
Question: You will often return to nucleophilic substitution, in particular the SN2 reaction, in subsequent chapters and concentrate on the nucleophile rather than the alkyl halide. By using different nucleophiles, nucleophilic substitution allows the synthesis of a wide variety of organic compounds with many different functional groups. With this in mind, draw the products of each two-step sequence. (Hint: Step [1] in each part involves an acid-base reaction that removes the most acidic hydrogen from the starting material.)
a.
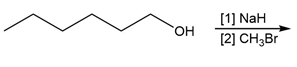
b.
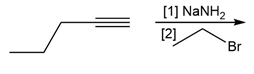
c.
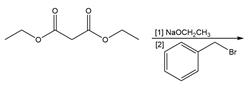
What do you think about this solution?
We value your feedback to improve our textbook solutions.