Chapter 8: Q.6 (page 303)
Which alkene in each pair is more stable?

a.

b.

c.
Short Answer
a. In the case of a., the stable structure is:
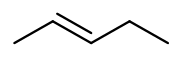
b. In the case of b., the stable structure is:
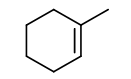
c. In the case of c., the stable structure is:
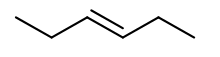
Learning Materials
Features
Discover
Chapter 8: Q.6 (page 303)
Which alkene in each pair is more stable?

a.

b.

c.
a. In the case of a., the stable structure is:

b. In the case of b., the stable structure is:

c. In the case of c., the stable structure is:

All the tools & learning materials you need for study success - in one app.
Get started for free
Consider an E2 reaction between and . What effect does each of the following changes have on the rate of elimination? (a) The base is changed to KOH. (b) The alkylhalide is changed to .
Classify each alkene in the following vitamins by the number of carbon substituents bonded to the double bond.
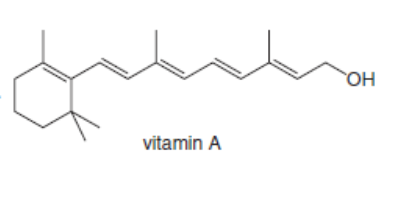
a.
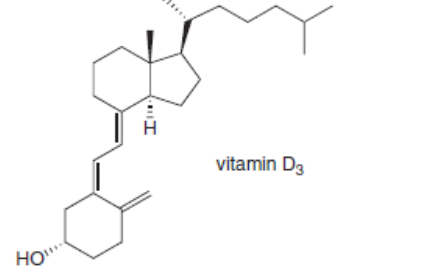
b.
For which reaction mechanisms—,E1, or E2—are each of the following statements true? A statement may be true for one or more mechanisms.
a. The mechanism involves carbocation intermediates.
b. The mechanism has two steps.
c. The reaction rate increases with better leaving groups.
d. The reaction rate increases when the solvent is changed from to localid="1648646954099" .
e. The reaction rate depends on the concentration of the alkyl halide only.
f. The mechanism is concerted.
g. The reaction of localid="1648646960420" with NaOH occurs by this mechanism.
h. Racemization at a stereogenic center occurs.
i. Tertiary (3°) alkyl halides react faster than 2° or 1° alkyl halides.
j. The reaction follows a second-order rate equation.
Which mechanism, E1 or E2, will occur in each reaction?
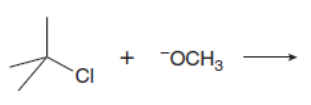
a.
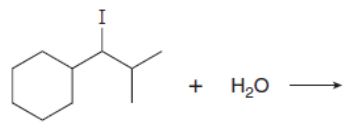
b.
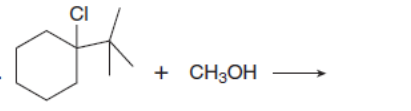
c.
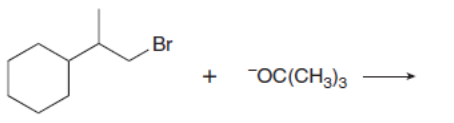
d.
Label the and carbons in each alkyl halide. Draw all possible elimination products formed when each alkyl halide is treated with role="math" localid="1648539176246" .
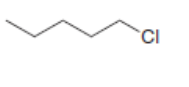
a.
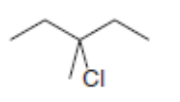
b.
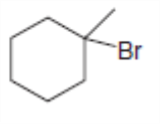
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.