Chapter 8: Q.59 (page 329)
Draw a stepwise, detailed mechanism for each reaction.
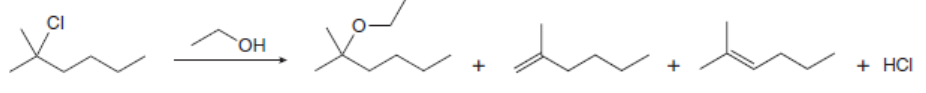
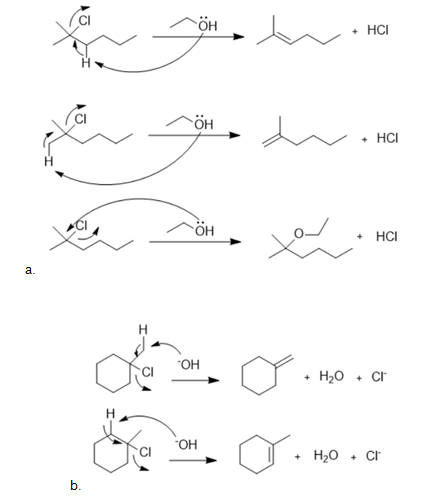
a.
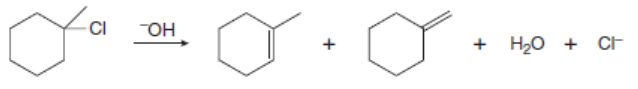
b.
Short Answer
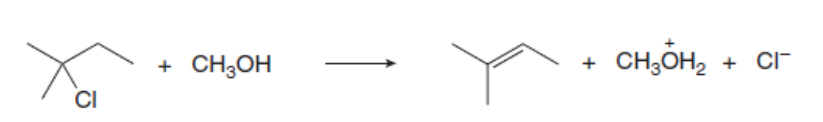
The mechanism for the formation of the substitution and elimination products are shown below:
Learning Materials
Features
Discover
Chapter 8: Q.59 (page 329)
Draw a stepwise, detailed mechanism for each reaction.
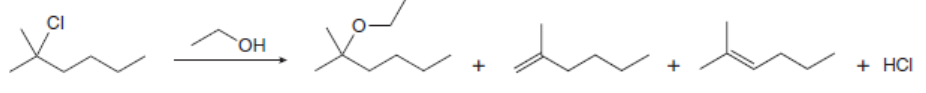
a.
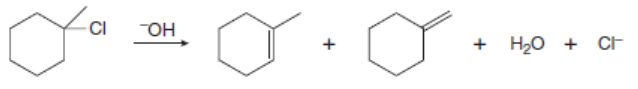
b.
The mechanism for the formation of the substitution and elimination products are shown below:

All the tools & learning materials you need for study success - in one app.
Get started for free
For which double bonds are stereoisomers possible?
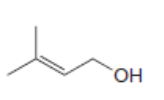
a.
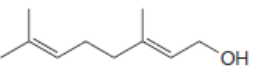
b.
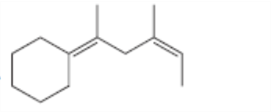
c.
How does each of the following changes affect the rate of an E1 reaction?
a. doubling
b. doubling
c. changing the halide from to
d. changing the leaving group from
e. changing the solvent from DMSO to
Several factors can affect alkene stability. Explain why alkene A is more stable than alkene B even though both contain disubstituted carbon-carbon double bonds.

Draw the alkynes formed when each dihalide is treated with excess base.
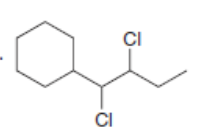
a.
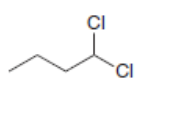
b.
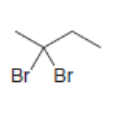
c.
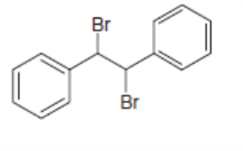
d.
Draw an E1 mechanism for the following reaction. Draw the structure of the transition state for each step.
What do you think about this solution?
We value your feedback to improve our textbook solutions.