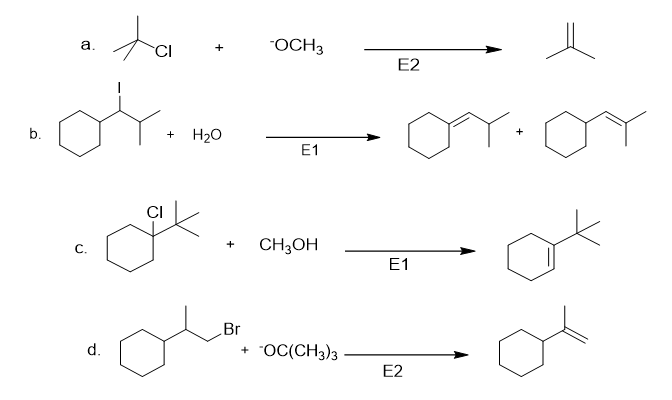
Chapter 8: Q.20 (page 318)
Which mechanism, E1 or E2, will occur in each reaction?
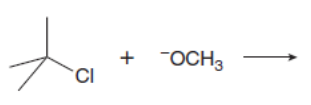
a.
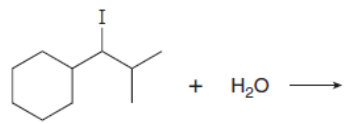
b.
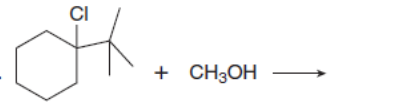
c.
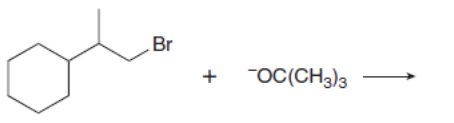
d.
Short Answer
The products with their mode of reaction are shown below:-
Learning Materials
Features
Discover
Chapter 8: Q.20 (page 318)
Which mechanism, E1 or E2, will occur in each reaction?
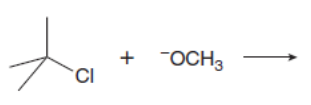
a.
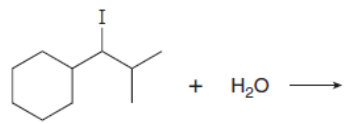
b.
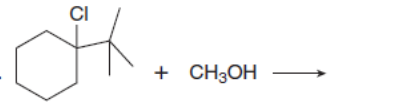
c.
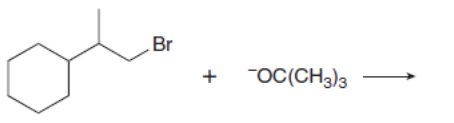
d.
The products with their mode of reaction are shown below:-

All the tools & learning materials you need for study success - in one app.
Get started for free
What is the major product formed when each alkyl halide is treated with each of the following reagents: [1] ; [2] ; [3]? If it is not possible to predict the major product, identify the products in the mixture and the mechanism by which each is formed.
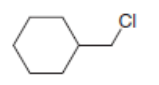
a.
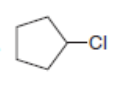
b.
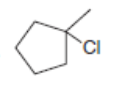
c.
Pick the reactant or solvent in each part that gives the faster elimination reaction.
a. reaction of with 1-chloro-1-methylcyclohexane or 1-chloro-3-methylcyclohexane
b. reaction of with or
c. reaction of with in or DMSO
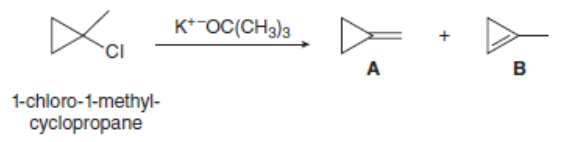
Dehydrohalogenation of 1-chloro-1-methyl cyclopropane affords two alkenes (A and B) as products. Explain why A is the major product despite the fact that it contains the less substituted double bond.
Draw the organic products formed in each reaction.
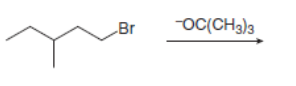
a.
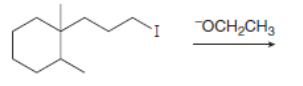
b.
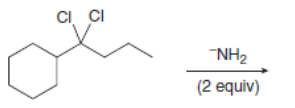
c.
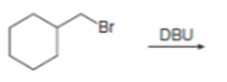
d.
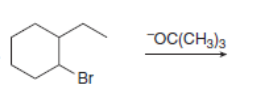
e.
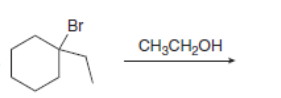
f.
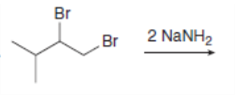
g.
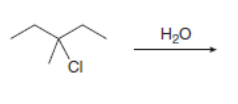
h.
Taking into account anti periplanar geometry, predict the major E2 product formed from each starting material.
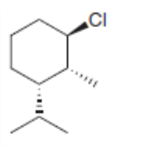
a.
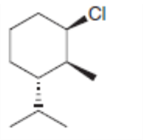
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.