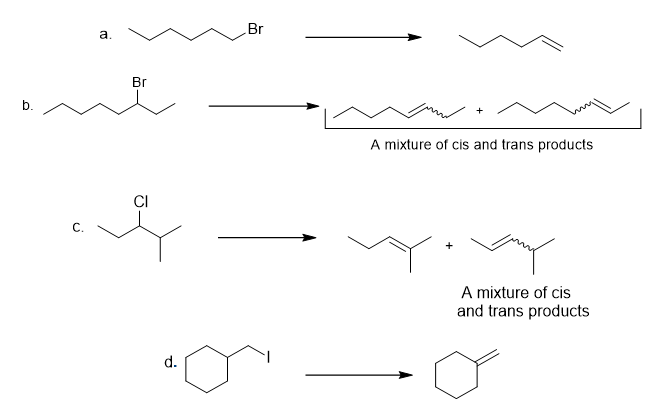
Chapter 8: Q.27 (page 325)
Draw all possible constitutional isomers formed by dehydrohalogenation of each alkyl halide.
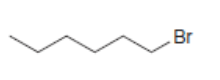
a.
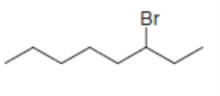
b.
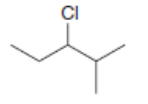
c.
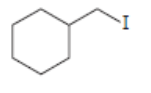
d.
Short Answer
All possible constitutional isomers formed by dehydrohalogenation of each alkyl halide are shown below:
Learning Materials
Features
Discover
Chapter 8: Q.27 (page 325)
Draw all possible constitutional isomers formed by dehydrohalogenation of each alkyl halide.

a.

b.

c.

d.
All possible constitutional isomers formed by dehydrohalogenation of each alkyl halide are shown below:

All the tools & learning materials you need for study success - in one app.
Get started for free
Under certain reaction conditions, 2,3-dibromobutane reacts with two equivalents of base to give three products, each of which contains two new bonds. Product A has two sp hybridized carbon atoms, product B has one sphybridized carbon atom, and product C has none. What are the structures of A, B, and C?
Rank the alkyl halides in order of increasing E2 reactivity. Then do the same for E1 reactivity
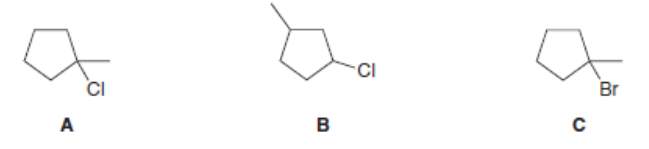
What is the major E2 elimination product formed from each halide?
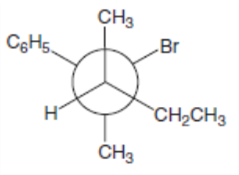
a.
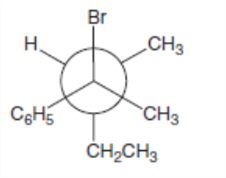
b.
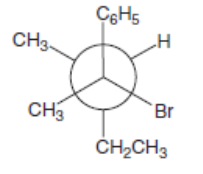
c.
Which mechanism, E1 or E2, will occur in each reaction?
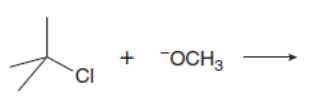
a.
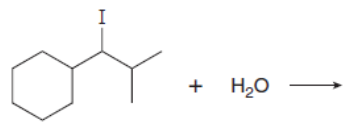
b.
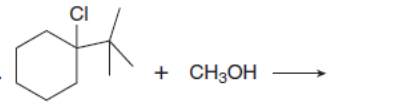
c.
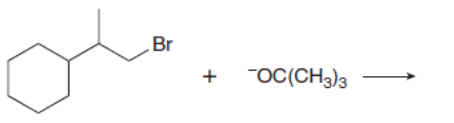
d.
Consider the following E2 reaction.
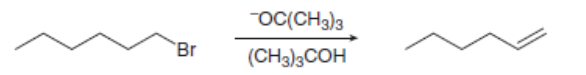
a. Draw the by-products of the reaction and use curved arrows to show the movement of
electrons.
b. What happens to the reaction rate with each of the following changes? [1] The solvent is changed to DMF. [2] The concentration of is decreased. [3] The base is changed to localid="1648628375469" . [4] The halide is changed to localid="1648628387551" [5] The leaving group is changed to localid="1648628394364" .
What do you think about this solution?
We value your feedback to improve our textbook solutions.