Chapter 8: Q.40 (page 327)
Rank the alkyl halides in order of increasing E2 reactivity. Then do the same for E1 reactivity
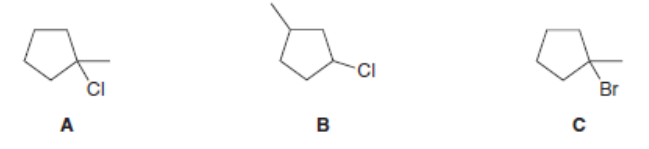
Short Answer
The given alkyl halides are ranked for E2 reactivity as shown:
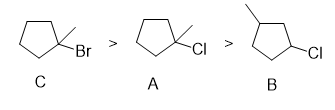
The same trend is followed for E1 too.

Learning Materials
Features
Discover
Chapter 8: Q.40 (page 327)
Rank the alkyl halides in order of increasing E2 reactivity. Then do the same for E1 reactivity

The given alkyl halides are ranked for E2 reactivity as shown:

The same trend is followed for E1 too.

All the tools & learning materials you need for study success - in one app.
Get started for free
Given that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each elimination. The alkyl halides in (a) and (b) are diastereomers of each other. How are the products of these two reactions related? Recall from Section 3.2A that is a phenyl group, a benzene ring bonded to another group.
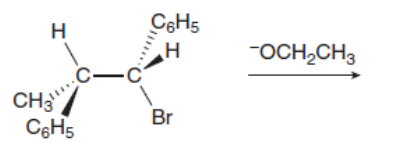
a.
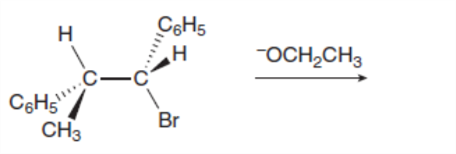
b.
(a) Which double bonds in (E)-ocimene, a major component of the odor of lilac flowers, can exhibit stereoisomerism? (b) Draw a diastereomer of (E)-ocimene

Draw all constitutional isomers formed in each E2 reaction and predict the major product using the Zaitsev rule.
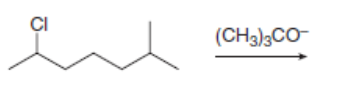
a.
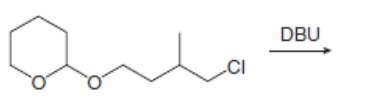
b.
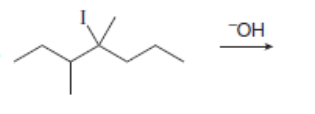
c.
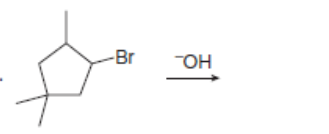
d.
For which double bonds are stereoisomers possible?
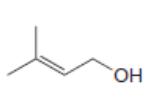
a.
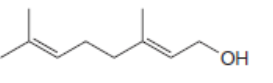
b.
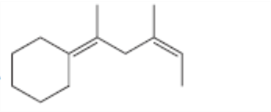
c.
Does cis-or trans-1-bromo-4-tert-butylcylohexane react faster in an E2 reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.