Chapter 8: Q.46 (page 328)
Does cis-or trans-1-bromo-4-tert-butylcylohexane react faster in an E2 reaction?
Short Answer
Cis-1-bromo-4-tert-butylcyclohexane reacts faster than trans-1-bromo-4-tert-butylcyclohexane in an E2 reaction.
Learning Materials
Features
Discover
Chapter 8: Q.46 (page 328)
Does cis-or trans-1-bromo-4-tert-butylcylohexane react faster in an E2 reaction?
Cis-1-bromo-4-tert-butylcyclohexane reacts faster than trans-1-bromo-4-tert-butylcyclohexane in an E2 reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw all constitutional isomers formed in each elimination reaction. Label the mechanism as E2 or E1.

a.
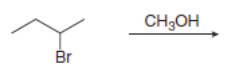
b.
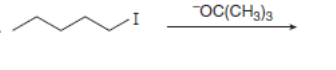
c.
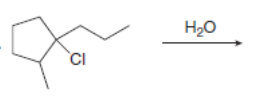
d.
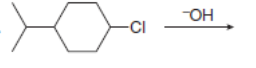
e.

f.
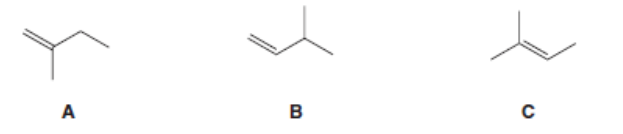
Rank the following alkenes in order of increasing stability:
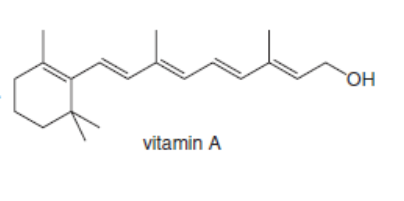
Classify each alkene in the following vitamins by the number of carbon substituents bonded to the double bond.
a.

b.
Draw a stepwise mechanism for the following reaction. The four-membered ring in the starting material and product is called aβ-lactam. This functional group confers biological activity on penicillin and many related antibiotics, as is discussed in Chapter 22. (Hint: The mechanism begins withβelimination and involves only two steps.)

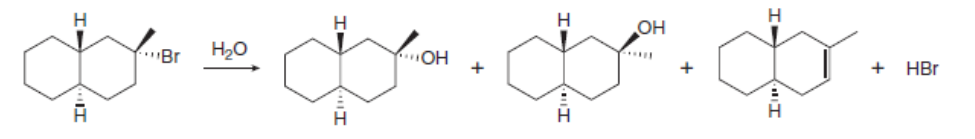
Draw a stepwise, detailed mechanism for the following reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.