Chapter 9: Q63. (page 379)
Question: Draw a stepwise, detailed mechanism for the following reaction.
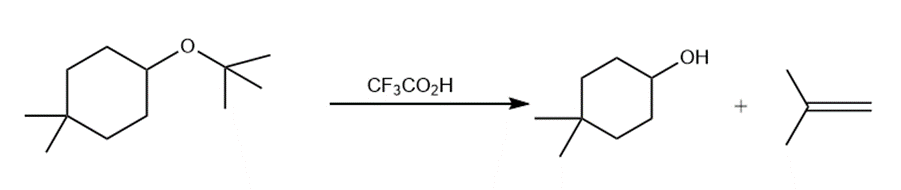
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 9: Q63. (page 379)
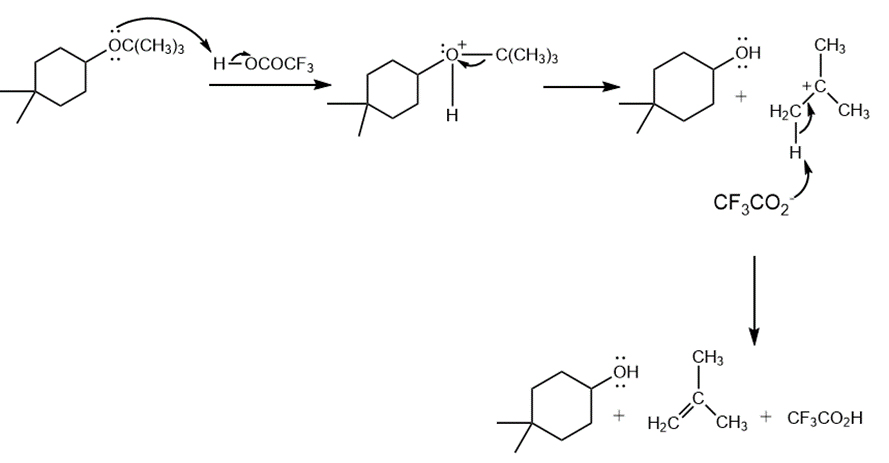
Question: Draw a stepwise, detailed mechanism for the following reaction.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
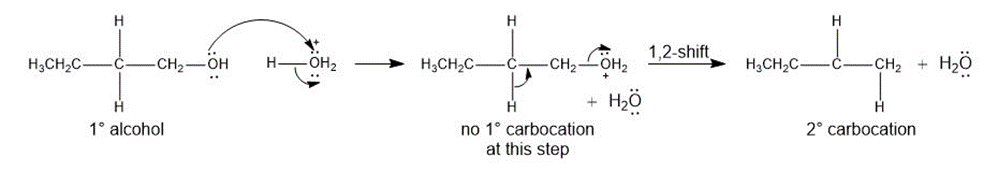
Question:Rearrangements can occur during the dehydration of alcohols even though no carbocation is formed-that is, a 1,2-shift occurs as the bond is broken, forming a more stable carbocation, as shown. Using this information, draw a stepwise mechanism that shows how is dehydrated with to form a mixture of and the cis and trans isomers of . We will see another example of this type of rearrangement in Section 18.5C.
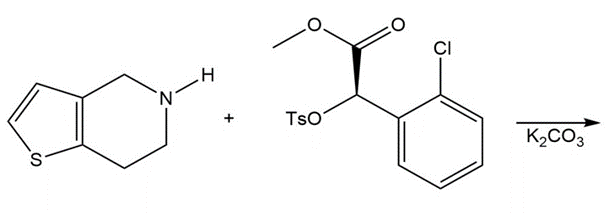
Question: Draw the product of the following reaction, one step in the synthesis of the antiplatelet agent clopidogrel used to reduce the risk of stokes.
Question: Rank the alcohols in order of increasing reactivity when dehydrated with
Question: Draw the products formed when ethylene oxide is treated with each reagent.
Question: Name each thiol.
a.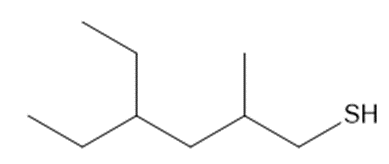
b.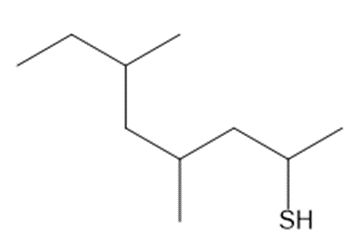
What do you think about this solution?
We value your feedback to improve our textbook solutions.