Chapter 2: Q. 2.65 (page 88)
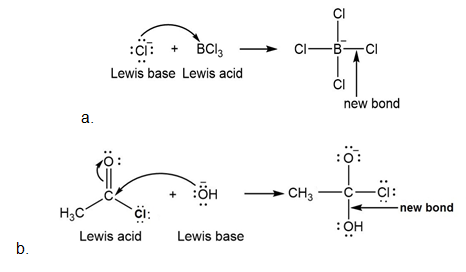
Label the Lewis acid and Lewis base in each reaction. Use curved arrows to show the movement of electron pairs.
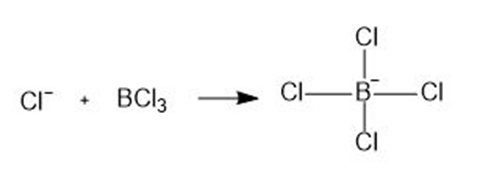
a.
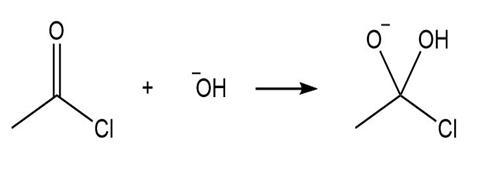
b.
Short Answer
Learning Materials
Features
Discover
Chapter 2: Q. 2.65 (page 88)
Label the Lewis acid and Lewis base in each reaction. Use curved arrows to show the movement of electron pairs.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the products formed when propan-2-ol the main ingredient in rubbing alcohol, is treated with each acid or base: a. NaH b. c. d.
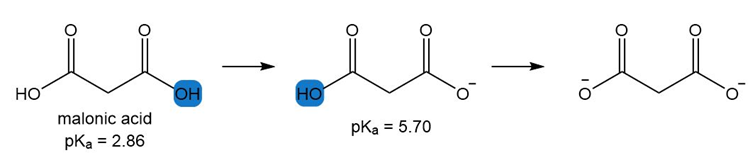
Two values are reported for malonic acid, a compound with two COOH groups. Explain why one is lower and one is higher than the of acetic acid
Draw the products of each acid-base reaction.
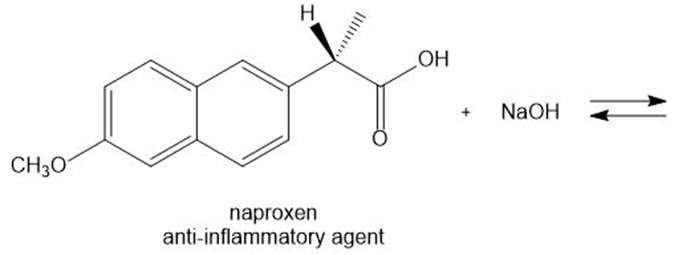
a.
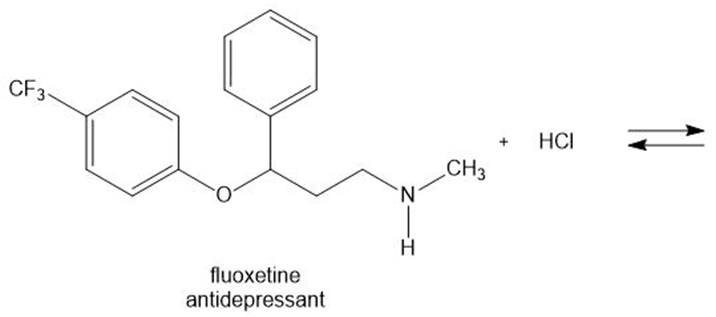
b.
Amino acids such as glycine are the building blocks of large molecules called proteins that give structure to muscle, tendon, hair, and nails.
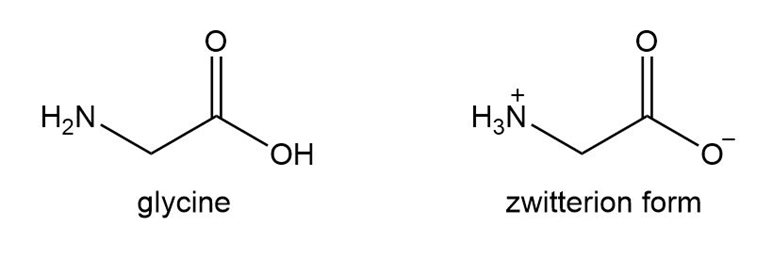
a. Explain why glycine does not actually exist in the form with all atoms uncharged, but actually exists as a salt called a zwitterion.
b. What product is formed when glycine is treated with concentrated HCl?
c. What product is formed when glycine is treated with NaOH?
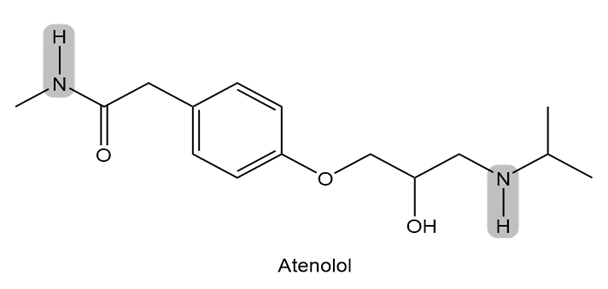
Atenolol is a (beta) blocker, a drug used to treat high blood pressure. Which of the indicated N-H bonds is more acidic? Explain your reasoning.
What do you think about this solution?
We value your feedback to improve our textbook solutions.