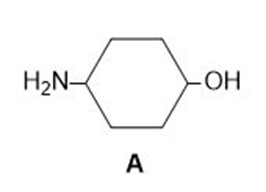
Chapter 2: Q. 2.55 (page 87)
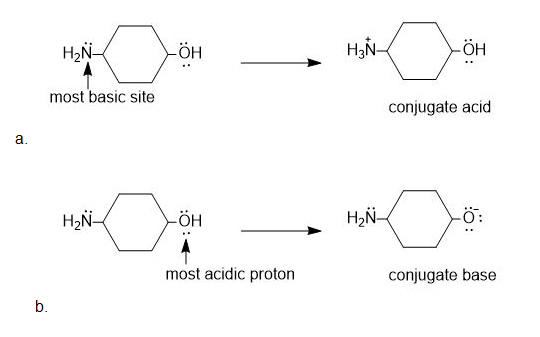
a. What is the conjugate acid of A?
b. What is the conjugate base of A?
Short Answer
Expert verified
Learning Materials
Features
Discover
Chapter 2: Q. 2.55 (page 87)
a. What is the conjugate acid of A?
b. What is the conjugate base of A?


All the tools & learning materials you need for study success - in one app.
Get started for free
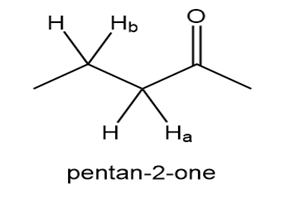
Explain why the bond is much more acidic than the bond in pentan-2-one.
What is the conjugate acid of each base?
a.
b.
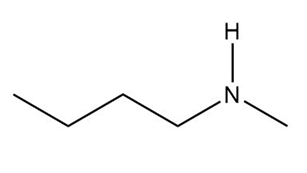
c.
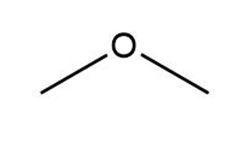
d.
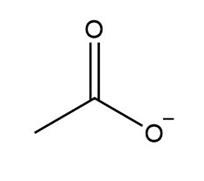
Which compounds can be deprotonated by , so that equilibrium favors the products? Refer to the pKa table in Appendix A.
a.HCOOH
b.
c.
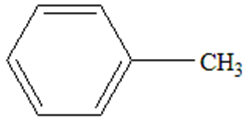
d.
Draw the products of each Lewis acid-base reaction. Label the electrophile and nucleophile.
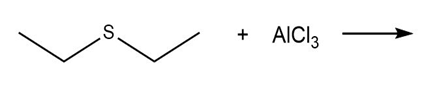
a.
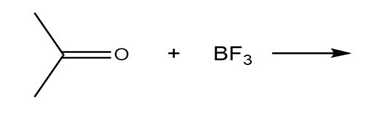
b.
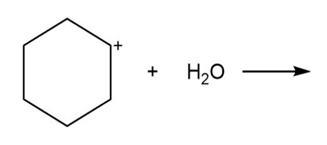
c.
What is for each compound? Use a calculator when necessary.
a.
b.
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.