Chapter 2: Q. 2.51 (page 87)
Rank the following ions in the order of increasing basicity:
a.
b.
c.
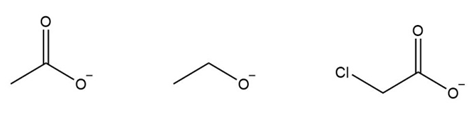
d.

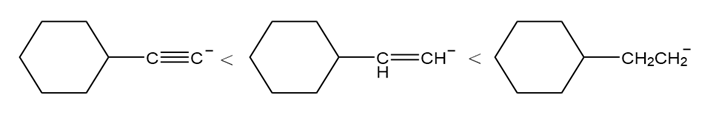
Short Answer
a.
b.
c.
d.
Learning Materials
Features
Discover
Chapter 2: Q. 2.51 (page 87)
Rank the following ions in the order of increasing basicity:
a.
b.
c.

d.

a.
b.
c.
d.

All the tools & learning materials you need for study success - in one app.
Get started for free
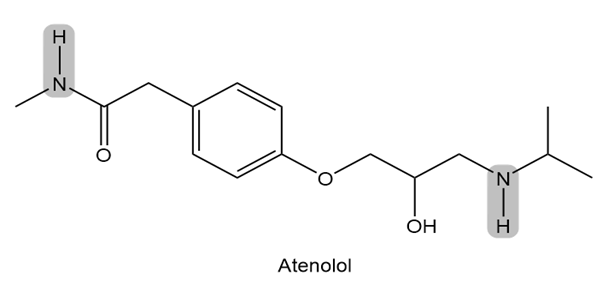
Atenolol is a (beta) blocker, a drug used to treat high blood pressure. Which of the indicated N-H bonds is more acidic? Explain your reasoning.
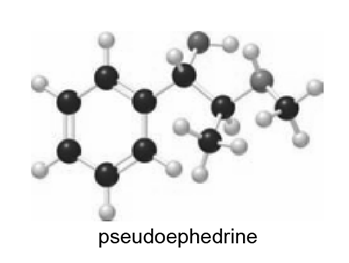
Which hydrogen in pseudoephedrine, the nasal decongestant in the commercial medication Sudafed, is most acidic?
Which of the following bases are strong enough to deprotonate ( = 25), so that equilibrium favors the products:
Which hydrogen in each molecule is most acidic?
a.
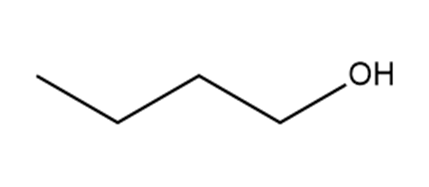
b.
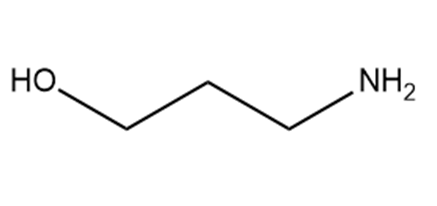
c.
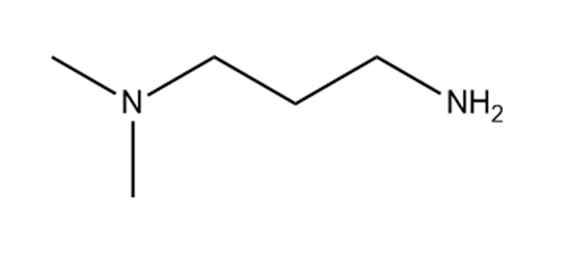
Amphetamine is a powerful stimulant of the central nervous system.

a. Which proton in amphetamine is most acidic? b. What products are formed when amphetamine is treated with NaH? c. What products are formed when amphetamine is treated with HCl?
What do you think about this solution?
We value your feedback to improve our textbook solutions.