Chapter 2: Q. 2.50 (page 86)
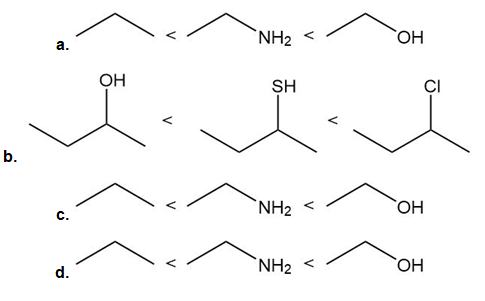
Rank the following compounds in order of increasing acidity.
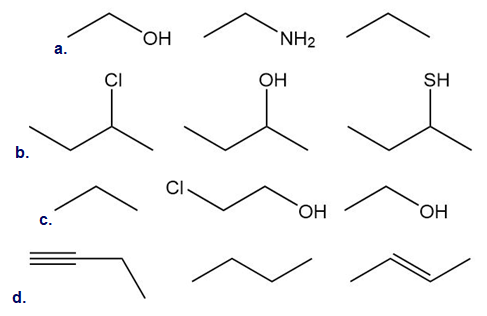
Short Answer
Learning Materials
Features
Discover
Chapter 2: Q. 2.50 (page 86)
Rank the following compounds in order of increasing acidity.


All the tools & learning materials you need for study success - in one app.
Get started for free
Many drugs are Brønsted-Lowry acids or bases.
a. What is the most acidic proton in the analgesic ibuprofen? Draw the conjugate base.
b. What is the most basic electron pair in cocaine? Draw the conjugate acid.

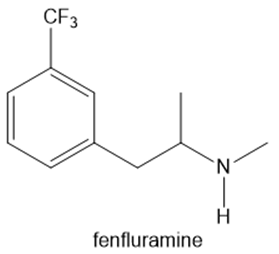
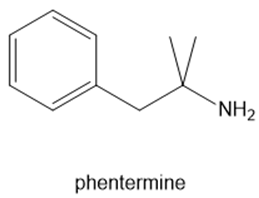
Fenfluramine and phentermine are two components of fen–phen, an appetite suppressant withdrawn from the market in 1997 after it was shown to damage the heart valves in some patients. What products are formed when fenfluramine and phentermine are each treated with acetic acid ?
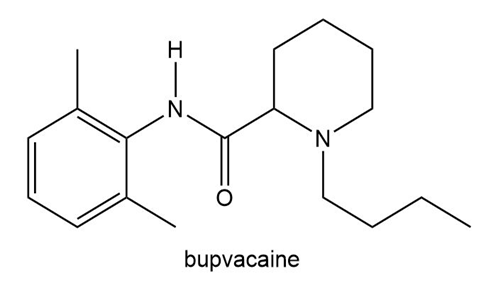
Bupivacaine (trade name Marcaine) is a quick-acting anesthetic often used during labor and delivery. Which nitrogen atom in bupivacaine is more basic? Explain your reasoning.
Draw the products formed when propan-2-ol the main ingredient in rubbing alcohol, is treated with each acid or base: a. NaH b. c. d.
Write a stepwise reaction sequence using proton transfer reactions to show how the following reaction occurs. (Hint: As a first step, use to remove a proton from the group between the C=O and C=C.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.