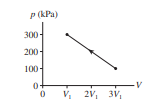
Chapter 19: Q. 3 (page 543)
80 J of work are done on the gas in the process shown in Figure EX19.3. What is V1 in cm3 ?
Short Answer
The value of is
Learning Materials
Features
Discover
Chapter 19: Q. 3 (page 543)
80 J of work are done on the gas in the process shown in Figure EX19.3. What is V1 in cm3 ?

The value of is
All the tools & learning materials you need for study success - in one app.
Get started for free
You come into the lab one day and find a well-insulated thermos bottle containing of boiling liquid nitrogen. The remainder of the thermos has nitrogen gas at a pressure of atm. The gas and liquid are in thermal equilibrium. While waiting for lab to start, you notice a piece of iron on the table with " " written on it. Just for fun, you drop the iron into the thermos and seal the cap tightly so that no gas can escape. After a few seconds have passed, what is the pressure inside the thermos? The density of liquid nitrogen is .
Two containers of a diatomic gas have the same initial conditions. One container, heated at constant pressure, has a temperature increase of . The other container receives the same quantity of heat energy, but at constant volume. What is its temperature increase?
You are boiling pasta and absentmindedly grab a copper stirring spoon rather than your wooden spoon. The copper spoon has a 20 mm 1.5 mm rectangular cross section, and the distance from the boiling water to your 35°C hand is 18 cm. How long does it take the spoon to transfer 25 J of energy to your hand?
III FIGURE CP19.79 shows a thermodynamic process followed by of hydrogen. How much heat energy is transferred to the gas?
52. An ideal-gas process is described by , where is a CALC constant.
a. Find an expression for the work done on the gas in this process as the volume changes from to .
b. of gas at an initial temperature of is compressed, using this process, from to . How much work is done on the gas?
c. What is the final temperature of the gas in ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.