Chapter 19: Q. 79 (page 547)
III FIGURE CP19.79 shows a thermodynamic process followed by of hydrogen. How much heat energy is transferred to the gas?
Short Answer
The amount of heat energy transferred to the gas is
Learning Materials
Features
Discover
Chapter 19: Q. 79 (page 547)
III FIGURE CP19.79 shows a thermodynamic process followed by of hydrogen. How much heat energy is transferred to the gas?
The amount of heat energy transferred to the gas is
All the tools & learning materials you need for study success - in one app.
Get started for free
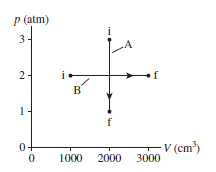
Two cylinders each contain of a diatomic gas at and a pressure of . Cylinder A expands isothermally and cylinder B expands adiabatically until the pressure of each is .
a. What are the final temperature and volume of each?
b. Show both processes on a single diagram. Use an appropriate scale on both axes.
A 65 cm3 block of iron is removed from an 800C furnace and
immediately dropped into 200 mL of 20C water. What fraction
of the water boils away?
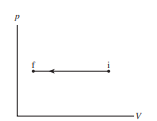
Draw a first-law bar chart (see Figure 19.12) for the gas process in Figure EX19.6.
A typical nuclear reactor generates 1000 MW (1000 MJ/s) of electrical energy. In doing so, it produces 2000 MW of “waste heat” that must be removed from the reactor to keep it from melting down. Many reactors are sited next to large bodies of water so that they can use the water for cooling. Consider a reactor where the intake water is at 18°C. State regulations limit the temperature of the output water to 30°C so as not to harm aquatic organisms. How many liters of cooling water have to be pumped through the reactor each minute?
0.10 mol of nitrogen gas follow the two processes shown in
Figure EX19.31. How much heat is required for each?
What do you think about this solution?
We value your feedback to improve our textbook solutions.