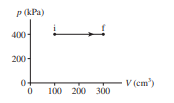
Chapter 19: Q. 63 (page 546)
Two containers of a diatomic gas have the same initial conditions. One container, heated at constant pressure, has a temperature increase of . The other container receives the same quantity of heat energy, but at constant volume. What is its temperature increase?
Short Answer
The temperature increase ator