Chapter 19: Q. 1 (page 543)
How much work is done on the gas in the process shown in Figure EX19.1?
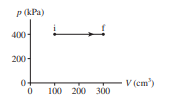
Short Answer
The amount of work done energy is
Learning Materials
Features
Discover
Chapter 19: Q. 1 (page 543)
How much work is done on the gas in the process shown in Figure EX19.1?

The amount of work done energy is
All the tools & learning materials you need for study success - in one app.
Get started for free
of a diatomic gas, with initial temperature , are compressed from to in a process in which constant. How much heat energy is added during this process?
The volume of a gas is halved during an adiabatic compression
that increases the pressure by a factor of 2.5.
a. What is the specific heat ratio?
b. By what factor does the temperature increase?
A cylindrical copper rod and an iron rod with exactly the same dimensions are welded together end to end. The outside end of the copper rod is held at , and the outside end of the iron rod is held at . What is the temperature at the midpoint where the rods are joined together?
A 500 g metal sphere is heated to 300 C, then dropped into a
beaker containing 300 cm3 of mercury at 20.0 C. A short time later
the mercury temperature stabilizes at 99.0 C. Identify the metal.
You need to raise the temperature of a gas by 10°C. To use the least amount of heat energy, should you heat the gas at constant pressure or at constant volume? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.