Chapter 39: Q 8 Exercise (page 1136)
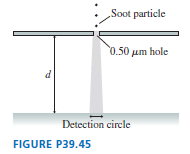
1.0x 1010 photons pass through an experimental apparatus. How many of them land in a 0.10-mm-wide strip where the probability density is 20 m-1?
Short Answer
Thus, the number of photons that will land in 0.10 mm wide strip is