Chapter 39: Q. 51 (page 1138)
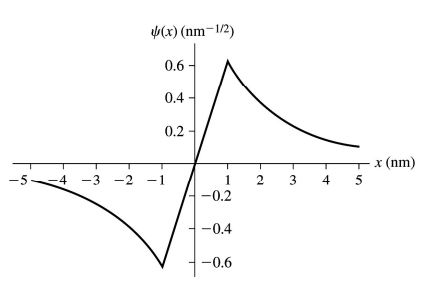
Consider the electron wave function
where x is in nm.
a. Determine the normalization constant c.
b. Draw a graph of over the interval role="math" localid="1650907186096" .
Provide numerical scales on both axes.
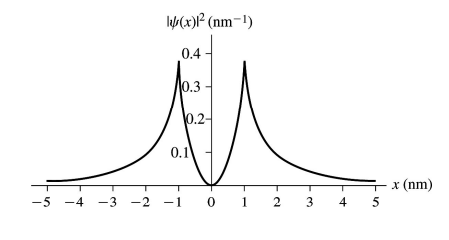
c. Draw a graph of over the interval role="math" localid="1650907657944" .
Provide numerical scales.
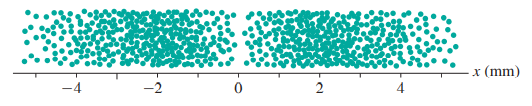
d. If electrons are detected, how many will be in the interval
role="math" localid="1650908765290" ?
Short Answer
a. The value of normalization constant is
c. The number of electrons are detected in the given interval is