Chapter 39: Q. 45 (page 1139)
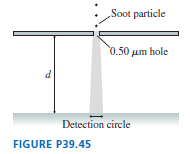
Soot particles, from incomplete combustion in diesel engines, are typically in diameter and have a density of . FIGURE P39.45 shows soot particles released from rest, in vacuum, just above a thin plate with a -diameter holeroughly the wavelength of visible light. After passing through the hole, the particles fall distance d and land on a detector. If soot particles were purely classical, they would fall straight down and, ideally, all land in a -diameter circle. Allowing for some experimental imperfections, any quantum effects would be noticeable if the circle diameter were . How far would the particles have to fall to fill a circle of this diameter?
Short Answer
The particles would have to fall through d=50 to notice quantum effects.


 FIGURE P39.46
FIGURE P39.46