Chapter 39: Q.46 - Excercises And Problems (page 1118)
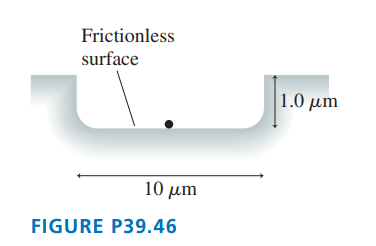
A small speck of dust with mass has fallen into the hole shown in FIGURE P39.46 and appears to be at rest. According to the uncertainty principle, could this particle have enough energy to get out of the hole? If not, what is the deepest hole of this width from which it would have a good chance to escape?
Short Answer
The value of less than the value of , so the particle will have enough energy to get out the hole. The deepest hole is for which, particle have a good chance to escape from the hole..

