Chapter 39: Q.25 (page 1137)
What is the minimum uncertainty in position, in , of an electron whose velocity is known to be between and ?
Short Answer
Round off to two significant figures, the minimum uncertainty in position of the electron is
Learning Materials
Features
Discover
Chapter 39: Q.25 (page 1137)
What is the minimum uncertainty in position, in , of an electron whose velocity is known to be between and ?
Round off to two significant figures, the minimum uncertainty in position of the electron is
All the tools & learning materials you need for study success - in one app.
Get started for free
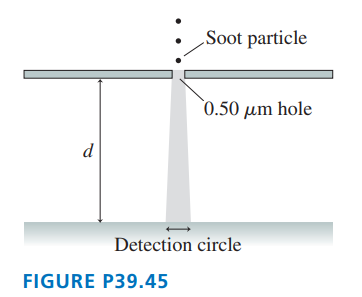
Soot particles, from incomplete combustion in diesel engines, are typically in diameter and have a density of . FIGURE P39.45 shows soot particles released from rest, in vacuum, just above a thin plate with a -diameter holeroughly the wavelength of visible light. After passing through the hole, the particles fall distance and land on a detector. If soot particles were purely classical, they would fall straight down and, ideally, all land in a -diameter circle. Allowing for some experimental imperfections, any quantum effects would be noticeable if the circle diameter were . How far would the particles have to fall to fill a circle of this diameter?
shows the probability density for an electron that has passed through an experimental apparatus. What is the probability that the electron will land in a
Suppose you draw a card from a regular deck of 52 cards.
a. What is the probability that you draw an ace?
b. What is the probability that you draw a spade?
The wave function of a particle is
where b is a positive constant. Find the probability that the particle is located in the interval -bx b
Consider a single-slit diffraction experiment using electrons. (Single-slit diffraction was described in Section 33.4.) Using Figure 39.5 as a model, draw
a. A dot picture showing the arrival positions of the first 40 or 50 electrons.
b. A graph of for the electrons on the detection screen.
c. A graph of for the electrons. Keep in mind that , as a wave-like function, oscillates between positive and negative.
What do you think about this solution?
We value your feedback to improve our textbook solutions.