Chapter 38: Q. 21 (page 1114)
The diameter of the nucleus is about . What is the kinetic energy, in MeV, of a proton with a de Broglie wavelength of ?
Learning Materials
Features
Discover
Chapter 38: Q. 21 (page 1114)
The diameter of the nucleus is about . What is the kinetic energy, in MeV, of a proton with a de Broglie wavelength of ?
All the tools & learning materials you need for study success - in one app.
Get started for free
a. What wavelength photon does a hydrogen atom emit in a transition?
b. What is the difference in the wavelengths emitted in a localid="1650823513478" transition and a transition?
Starting from Equation 38.32, derive Equation 38.33
I FIGURE EX38.24 is an energy-level diagram for a simple atom. What wavelengths, in \mathrm{nm}, appear in the atom's (a) emission spectrum and (b) absorption spectrum?
FIGURE EX38.24 n=1-E_{1}=0.00 \mathrm{eV}
A incandescent lightbulb emits about of visible light. (The other are emitted as infrared radiation or lost as heat to the surroundings.) The average wavelength of the visible light is about , so make the simplifying assumption that all the light has this wavelength. How many visible-light photons does the bulb emit per second?
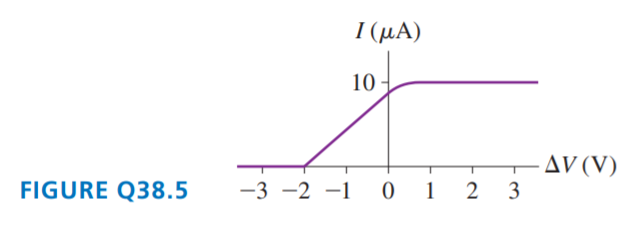
FIGURE Q38.5 is the current-versus-potential-difference graph for a photoelectric-effect experiment with an unknown metal. If classical physics provided the correct description of the photoelectric effect, how would the graph look if:
a. The light was replaced by an equally intense light with a shorter wavelength? Draw it.
b. The metal was replaced by a different metal with a smaller work function? Draw it.
What do you think about this solution?
We value your feedback to improve our textbook solutions.