Chapter 38: Q. 58 (page 1116)
Starting from Equation 38.32, derive Equation 38.33
Short Answer
The Derive equation is
Learning Materials
Features
Discover
Chapter 38: Q. 58 (page 1116)
Starting from Equation 38.32, derive Equation 38.33
The Derive equation is
All the tools & learning materials you need for study success - in one app.
Get started for free
Very large, hot stars—much hotter than our sun—can be identified by the way in which ions in their atmosphere absorb light. What are the three longest wavelengths, in nm, in the Balmer series of ?
a. Determine the energy, in eV, of a photon with a 550 nm wavelength.
b. Determine the wavelength of a 7.5 keV x-ray photon.
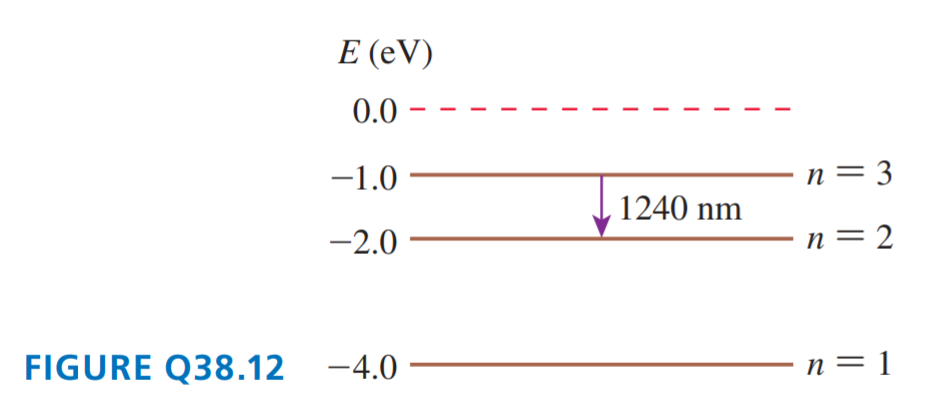
FIGURE Q38.12 shows the energy-level diagram of Element X.
a. What is the ionization energy of Element X?
b. An atom in the ground state absorbs a photon, then emits a photon with a wavelength of 1240 nm. What was the energy of the photon that was absorbed?
c. An atom in the ground state has a collision with an electron, then emits a photon with a wavelength of 1240 nm. What conclusion can you draw about the initial kinetic energy of the electron?
Which metals in Table 38.1 exhibit the photoelectric effect for (a) light with and (b) light with ?
Through what potential difference must an electron be accelerated from rest to have a de Broglie wavelength of ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.