Chapter 38: Q. 18 (page 1114)
Through what potential difference must an electron be accelerated from rest to have a de Broglie wavelength of ?
Short Answer
The potential difference
Learning Materials
Features
Discover
Chapter 38: Q. 18 (page 1114)
Through what potential difference must an electron be accelerated from rest to have a de Broglie wavelength of ?
The potential difference
All the tools & learning materials you need for study success - in one app.
Get started for free
I What is the radius of a hydrogen atom whose electron is bound by ?
The charge on a muon-a subatomic particle is -e and its mass 207 times that of an electron-
which is confined in a 15-pm-long, one-dimensional box.
(1 pm = 1 picometer = 10-12 m.)
What is the wavelength, in nm, of the photon emitted in a quantum jump from n=2 to n=1
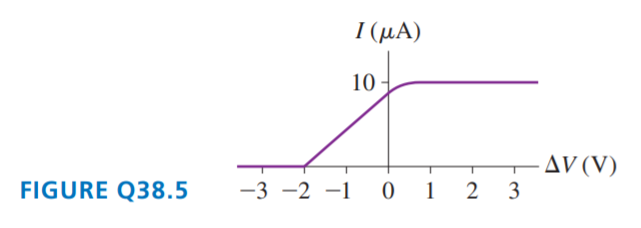
FIGURE Q38.5 is the current-versus-potential-difference graph for a photoelectric-effect experiment with an unknown metal. If classical physics provided the correct description of the photoelectric effect, how would the graph look if:
a. The light was replaced by an equally intense light with a shorter wavelength? Draw it.
b. The metal was replaced by a different metal with a smaller work function? Draw it.
\| An electron with 2.00 mathrm{eV of kinetic energy collides with the atom shown in FIGURE EX38.24.
a. Is the electron able to excite the atom? Why or why not?
b. If your answer to part a was yes, what is the electron's kinetic energy after the collision?
What lie the winding of the Irasuliens? , and in the hydronics ? In what spectral range do these lie?
What do you think about this solution?
We value your feedback to improve our textbook solutions.