Chapter 40: Q. 5 (page 1175)
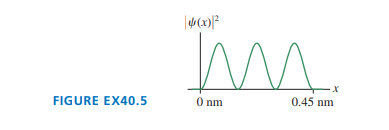
FIGURE EX is the probability density for an electron in a rigid box. What is the electron’s energy, in ?
Short Answer
The electron’s energy, in is.
Learning Materials
Features
Discover
Chapter 40: Q. 5 (page 1175)
FIGURE EX is the probability density for an electron in a rigid box. What is the electron’s energy, in ?

The electron’s energy, in is.
All the tools & learning materials you need for study success - in one app.
Get started for free
An electron is confined in a harmonic potential well that has a spring constant of 2.0 N/m. a. What are the first three energy levels of the electron? b. What wavelength photon is emitted if the electron undergoes a 3 S 1 quantum jump?
An electron approaches a wide potential-energy barrier of height . What energy electron has a tunneling probability of (a) 10%, (b) 1.0%, and (c) 0.10%?
Two adjacent energy levels of an electron in a harmonic potential well are known to be and . What is the spring constant of the potential well?
An electron confined in a harmonic potential well emits a 1200nm photon as it undergoes a quantum jump. What is the spring constant of the potential well?
Use the data from Figure 40.24 to calculate the first three vibrational energy levels of a carbon-oxygen double bond.
What do you think about this solution?
We value your feedback to improve our textbook solutions.