Chapter 40: Q. 24 (page 1175)
An electron approaches a wide potential-energy barrier of height . What energy electron has a tunneling probability of (a) 10%, (b) 1.0%, and (c) 0.10%?
Short Answer
a. The energy is
b.
c.
Learning Materials
Features
Discover
Chapter 40: Q. 24 (page 1175)
An electron approaches a wide potential-energy barrier of height . What energy electron has a tunneling probability of (a) 10%, (b) 1.0%, and (c) 0.10%?
a. The energy is
b.
c.
All the tools & learning materials you need for study success - in one app.
Get started for free
A helium atom is in a finite potential well. The atom’s energy isbelow . What is the atom’s penetration distance into the classically forbidden region?
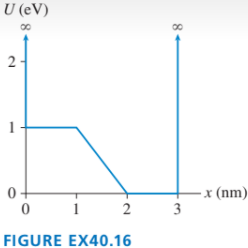
The graph in FIGURE EX40.16 shows the potential-energy function U(x of a particle. Solution of the Schrödinger equation finds that the n=3 level has and that the n=6 level has .
a. Redraw this figure and add to it the energy lines for the n=3 and n=6 states.
b. Sketch the n=3 and n=6 wave functions. Show them as oscillating about the appropriate energy line.
Model an atom as an electron in a rigid box of length , roughly twice the Bohr radius.
a. What are the four lowest energy levels of the electron?
b. Calculate all the wavelengths that would be seen in the emission spectrum of this atom due to quantum jumps between these four energy levels. Give each wavelength a label to indicate the transition.
c. Are these wavelengths in the infrared, visible, or ultraviolet portion of the spectrum?
d. The stationary states of the Bohr hydrogen atom have negative energies. The stationary states of this model of the atom have positive energies. Is this a physically significant difference? Explain.
e. Compare this model of an atom to the Bohr hydrogen atom. In what ways are the two models similar? Other than the signs of the energy levels, in what ways are they different?
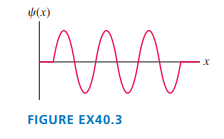
| FIGURE EX shows the wave function of an electron in a rigid box. The electron energy is. How long is the box?
The correspondence principle says that the average behavior of a quantum system should begin to look like the Newtonian solution in the limit that the quantum number becomes very large. What is meant by “the average behavior” of a quantum system?
What do you think about this solution?
We value your feedback to improve our textbook solutions.