Chapter 40: Q. 16 (page 1175)
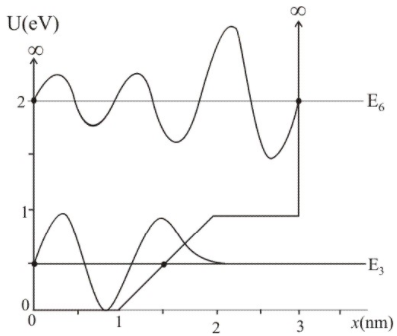
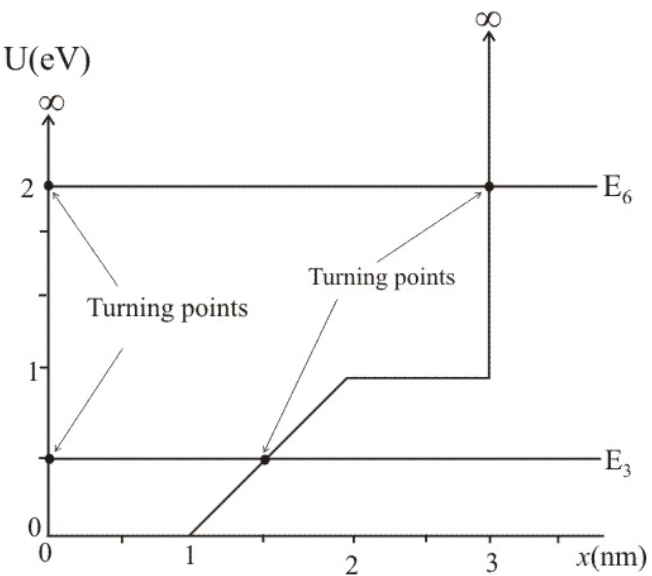
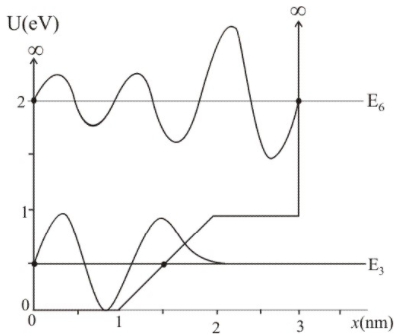
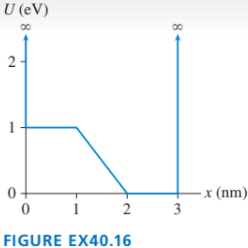
The graph in FIGURE EX40.16 shows the potential-energy function U(x of a particle. Solution of the Schrödinger equation finds that the n=3 level has and that the n=6 level has .
a. Redraw this figure and add to it the energy lines for the n=3 and n=6 states.
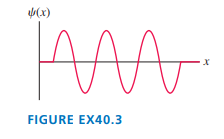
b. Sketch the n=3 and n=6 wave functions. Show them as oscillating about the appropriate energy line.
Short Answer
(a) The 3 energy level of energy 0.5eV and 6 energy level of energy 2.30eV is shown in the figure.
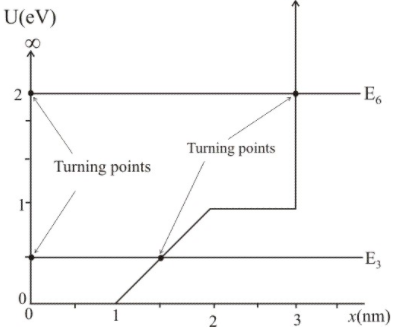
(b)