Chapter 40: Q. 5 (page 1174)
Rank in order, from largest to smallest, the penetration distancestoof the wave functions corresponding to the three energy levels in FIGURE Q.
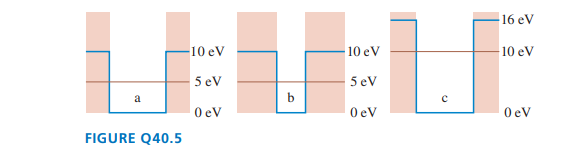
Short Answer
The wave functions corresponding to the three energy levels is
Learning Materials
Features
Discover
Chapter 40: Q. 5 (page 1174)
Rank in order, from largest to smallest, the penetration distancestoof the wave functions corresponding to the three energy levels in FIGURE Q.

The wave functions corresponding to the three energy levels is
All the tools & learning materials you need for study success - in one app.
Get started for free
Two adjacent energy levels of an electron in a harmonic potential well are known to be and . What is the spring constant of the potential well?
A particle in a potential well is in the quantum state. How many peaks are in the probability density ?
Consider a particle in a rigid box of length L. For each of the states and :
a. Sketch graphs of . Label the points and .
b. Where, in terms of L, are the positions at which the particle is most likely to be found?
c. Where, in terms of L, are the positions at which the particle is least likely to be found?
d. Determine, by examining your graphs, if the probability of finding the particle in the left one-third of the box is less than, equal to, or greater than . Explain your reasoning.
e. Calculate the probability that the particle will be found in the left one-third of the box
Sketch the n=1 and n=7 wave functions for the potential energy shown in FIGURE EX40.15
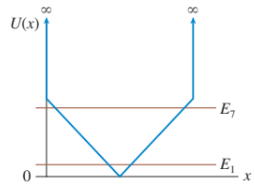
A particle of mass m has the wave function when it is in an allowed energy level with .
a. Draw a graph of versus.
b. At what value or values of is the particle most likely to be found?
c. Find and graph the potential-energy function .
What do you think about this solution?
We value your feedback to improve our textbook solutions.