Chapter 40: Q. 3 (page 1174)
A particle in a potential well is in the quantum state. How many peaks are in the probability density ?
Short Answer
There are five peaks in the probability density .
Learning Materials
Features
Discover
Chapter 40: Q. 3 (page 1174)
A particle in a potential well is in the quantum state. How many peaks are in the probability density ?
There are five peaks in the probability density .
All the tools & learning materials you need for study success - in one app.
Get started for free
The energy of an electron in a deep potential well is . At what distance into the classically forbidden region has the amplitude of the wave function decreased to of its value at the edge of the potential well?
A typical electron in a piece of metallic sodium has energycompared to a free electron, where is the work function of sodium.
a. At what distance beyond the surface of the metal is the electronŌĆÖs probability density of its value at the surface?
b. How does this distance compare to the size of an atom?
Figure 40.27a modeled a hydrogen atom as a finite potential well with rectangular edges. A more realistic model of a hydrogen atom, although still a one-dimensional model, would be the electron + proton electrostatic potential energy in one dimension:
a. Draw a graph of U(x) versus x. Center your graph at .
b. Despite the divergence at , the Schr├Čdinger equation can be solved to find energy levels and wave functions for the electron in this potential. Draw a horizontal line across your graph of part a about one-third of the way from the bottom to the top. Label this line , then, on this line, sketch a plausible graph of the wave function.
c. Redraw your graph of part a and add a horizontal line about two-thirds of the way from the bottom to the top. Label this line , then, on this line, sketch a plausible graph of the wave function.
Even the smoothest mirror finishes are ŌĆ£roughŌĆØ when viewed at a scale of . When two very smooth metals are placed in contact with each other, the actual distance between the surfaces varies fromat a few points of real contact to . The average distance between the surfaces is. The work function of aluminum is . What is the probability that an electron will tunnel between two pieces of aluminum that are apart? Give your answer as a power ofrather than a power of.
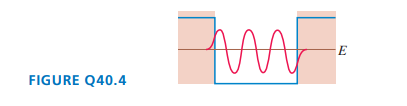
What is the quantum number of the particle in FIGURE Q? How can you tell?
What do you think about this solution?
We value your feedback to improve our textbook solutions.