Chapter 40: Q. 4 (page 1174)
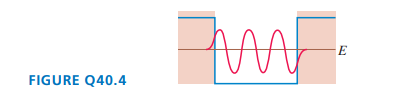
What is the quantum number of the particle in FIGURE Q? How can you tell?
Short Answer
The quantum number of the particle is.
Learning Materials
Features
Discover
Chapter 40: Q. 4 (page 1174)
What is the quantum number of the particle in FIGURE Q? How can you tell?

The quantum number of the particle is.
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider a quantum harmonic oscillator.
a. What happens to the spacing between the nodes of the wave function as |x| increases? Why?
b. What happens to the heights of the antinodes of the wave function as |x| increases? Why?
c. Sketch a reasonably accurate graph of the n=8 wave function of a quantum harmonic oscillator.
a. Derive an expression for the classical probability density for a ball that bounces between the ground and height. The collisions with the ground are perfectly elastic.
b. Graph your expression between .
c. Interpret your graph. Why is it shaped as it is?
For a particle in a finite potential well of width L and depth , what is the ratio of the probability Prob ( in at ) to the probability Prob ( in at)?
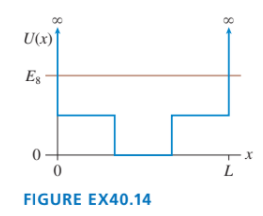
Sketch the wave function for the potential energy shown in FIGURE EX40.14.
Show that the constant b used in the quantum-harmonic-oscillator wave functions (a) has units of length and (b) is the classical turning point of an oscillator in the ground state.
What do you think about this solution?
We value your feedback to improve our textbook solutions.