Chapter 40: Q. 14 (page 1175)
Sketch the wave function for the potential energy shown in FIGURE EX40.14.
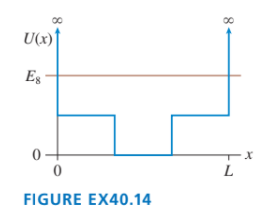
Short Answer
The shape of the n=8 wave function for the potential energy,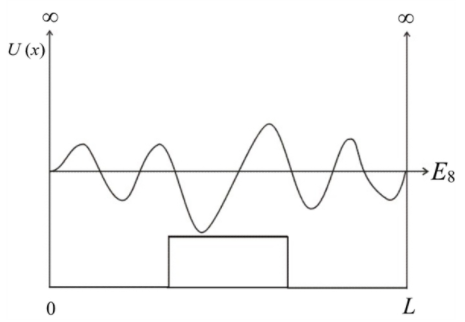
Learning Materials
Features
Discover
Chapter 40: Q. 14 (page 1175)
Sketch the wave function for the potential energy shown in FIGURE EX40.14.

The shape of the n=8 wave function for the potential energy,
All the tools & learning materials you need for study success - in one app.
Get started for free
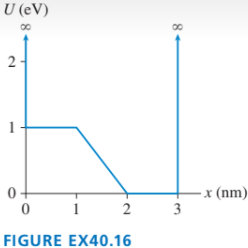
The graph in FIGURE EX40.16 shows the potential-energy function U(x of a particle. Solution of the Schrödinger equation finds that the n=3 level has and that the n=6 level has .
a. Redraw this figure and add to it the energy lines for the n=3 and n=6 states.
b. Sketch the n=3 and n=6 wave functions. Show them as oscillating about the appropriate energy line.
Two adjacent energy levels of an electron in a harmonic potential well are known to be and . What is the spring constant of the potential well?
The energy of an electron in a deep potential well is . At what distance into the classically forbidden region has the amplitude of the wave function decreased to of its value at the edge of the potential well?
A finite potential well has depth. What is the penetration distance for an electron with energy
(a)
(b) and
(c)?
Figure 40.27a modeled a hydrogen atom as a finite potential well with rectangular edges. A more realistic model of a hydrogen atom, although still a one-dimensional model, would be the electron + proton electrostatic potential energy in one dimension:
a. Draw a graph of U(x) versus x. Center your graph at .
b. Despite the divergence at , the Schrödinger equation can be solved to find energy levels and wave functions for the electron in this potential. Draw a horizontal line across your graph of part a about one-third of the way from the bottom to the top. Label this line , then, on this line, sketch a plausible graph of the wave function.
c. Redraw your graph of part a and add a horizontal line about two-thirds of the way from the bottom to the top. Label this line , then, on this line, sketch a plausible graph of the wave function.
What do you think about this solution?
We value your feedback to improve our textbook solutions.