Chapter 40: Q. 13 (page 1175)
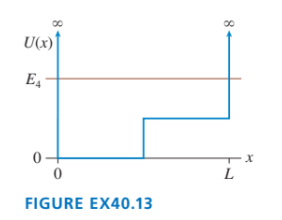
Sketch the n=4 wave function for the potential energy shown in FIGURE EX40.13.
Short Answer
The shape of the n=4 wave function for the potential energy,
Learning Materials
Features
Discover
Chapter 40: Q. 13 (page 1175)
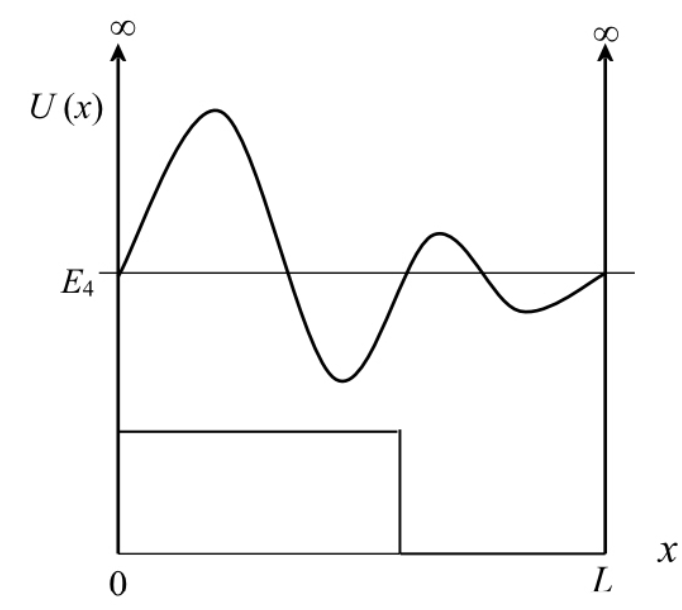
Sketch the n=4 wave function for the potential energy shown in FIGURE EX40.13.

The shape of the n=4 wave function for the potential energy,

All the tools & learning materials you need for study success - in one app.
Get started for free
Suppose that and are both solutions to the Schrödinger equation for the same potential energy . Prove that the superposition is also a solution to the Schrödinger equation.
a. Sketch graphs of the probability density for the four states in the finite potential well of Figure a. Stack them vertically, similar to the Figure a graph of .
b. What is the probability that a particle in the state of the finite potential well will be found at the center of the well? Explain.
c. Is your answer to part b consistent with what you know about standing waves? Explain.
a. Derive an expression for the classical probability density for a ball that bounces between the ground and height. The collisions with the ground are perfectly elastic.
b. Graph your expression between .
c. Interpret your graph. Why is it shaped as it is?
A -long box has a thin partition that divides the box into a-long section and a-long section. An electron confined in the shorter section is in the state. The partition is briefly withdrawn, then reinserted, leaving the electron in the longer section of the box. What is the electron’s quantum state after the partition is back in place?
a. Derive an expression for , the wavelength of light emitted by a particle in a rigid box during a quantum jump from
b. In what length rigid box will an electron undergoing a transition emit light with a wavelength of ? This is the wavelength of a ruby laser
What do you think about this solution?
We value your feedback to improve our textbook solutions.