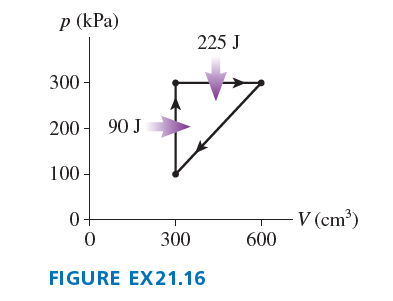
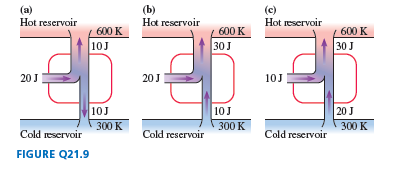
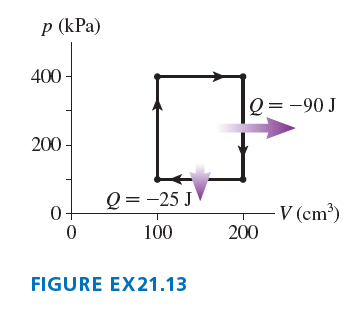
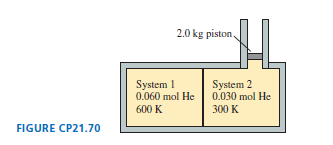
Chapter 21: Q.28 (page 595)
A Carnot engine whose hot-reservoir temperature is has a thermal efficiency of . By how many degrees should the temperature of the cold reservoir be decreased to raise the engine's efficiency to
Short Answer
The temperature decrease of the cold bath required is