Chapter 21: Q. 72 (page 599)
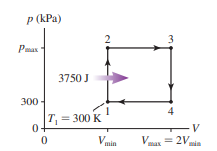
A heat engine using a diatomic ideal gas goes through the following closed cycle:
- Isothermal compression until the volume is halved.
- Isobaric expansion until the volume is restored to its initial value.
Isochoric cooling until the pressure is restored to its initial value. What are the thermal efficiencies of () this heat engine and
() a Carnot engine operating between the highest and lowest temperatures reached by this engine?
Short Answer
() The Thermal efficiency of the heat engine is
() The thermal efficiency of a Carnot engine operating between the highest and lowest temperatures reached by the engine is