Chapter 21: Q 3 (page 593)
Could you have a heat engine with ? Explain.
Short Answer
A heat engine withis No.
Learning Materials
Features
Discover
Chapter 21: Q 3 (page 593)
Could you have a heat engine with ? Explain.
A heat engine withis No.
All the tools & learning materials you need for study success - in one app.
Get started for free
A heat engine using of helium as the working substance follows the cycle shown in FIGURE P.
a. Determine the pressure, temperature, and volume of the gas at points ,, and .
b. What is the engine's thermal efficiency?
c. What is the maximum possible efficiency of a heat engine that operates between and?
A heat engine using 1.0 mol of a monatomic gas follows the cycle shown in FIGURE P21.55. 3750 J of heat energy is transferred to the gas during process 1 -> 2.
a. Determine Ws, Q, and ΔEth for each of the four processes in this cycle. Display your results in a table.
b. What is the thermal efficiency of this heat engine?
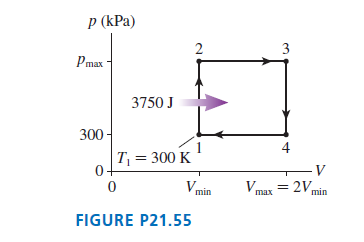
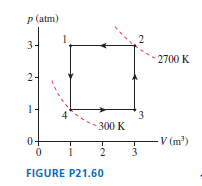
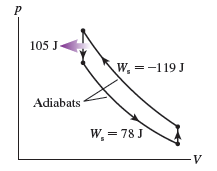
FIGURE Pis the diagram of Example , but now the device is operated in reverse.
a. During which processes is heat transferred into the gas?
b. Is this, heat extracted from a hot reservoir, or , heat extracted from a cold reservoir? Explain.
c. Determine the values ofand.
Hint: The calculations have been done in Example and do not need to be repeated. Instead, you need to determine which processes now contribute to and which to .
d. Is the area inside the curve or ? What is its value?
e. The device is now being operated in a ccw cycle. Is it a refrigerator? Explain.
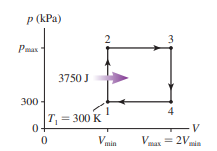
A heat engine using of a monatomic gas follows the cycle shown in FIGURE P. of heat energy is transferred to the gas during process .
a. Determine , and for each of the four processes in this cycle. Display your results in a table.
b. What is the thermal efficiency of this heat engine?
What are (a) the heat extracted from the cold reservoir and (b) the coefficient of performance for the refrigerator shown in Figure Ex-?
What do you think about this solution?
We value your feedback to improve our textbook solutions.