Chapter 21: Q. 38 (page 596)
Prove that the work done in an adiabatic process
Short Answer
We perform the integral for the work using the adiabatic process conditions to get
Learning Materials
Features
Discover
Chapter 21: Q. 38 (page 596)
Prove that the work done in an adiabatic process
We perform the integral for the work using the adiabatic process conditions to get
All the tools & learning materials you need for study success - in one app.
Get started for free
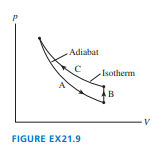
The cycle of FIGURE consists of three processes. Make a table with rows labeled and columns labeled, , and . Fill each box in the table with ,, or to indicate whether the quantity increases, decreases, or stays the same during that process.
The electric output of a power plant is . Cooling water flows through the power plant at the rate . The cooling water enters the plant at and exits at. What is the power plant’s thermal efficiency?
FIGURE Pshows the cycle for a heat engine that uses a gas having .The initial temperature is, and this engine operates atcycles per second.
a. What is the power output of the engine?
b. What is the engine's thermal efficiency?
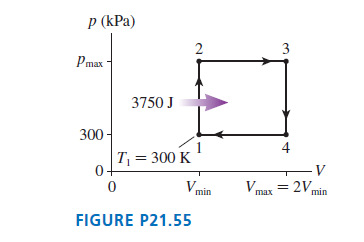
A heat engine using 1.0 mol of a monatomic gas follows the cycle shown in FIGURE P21.55. 3750 J of heat energy is transferred to the gas during process 1 -> 2.
a. Determine Ws, Q, and ΔEth for each of the four processes in this cycle. Display your results in a table.
b. What is the thermal efficiency of this heat engine?
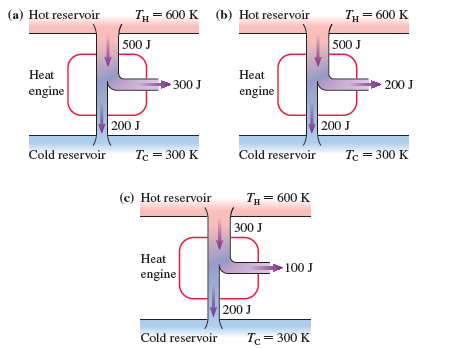
Which, if any, of the heat engines in FIGURE EX 21.22 violate (a) the first law of thermodynamics or (b) the second law of thermodynamics? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.